901488
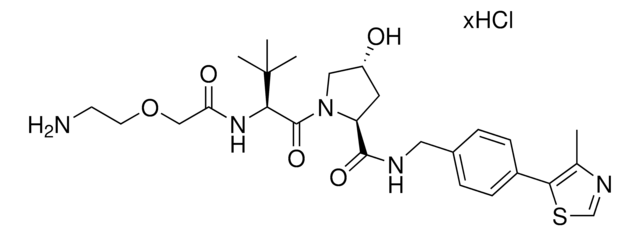
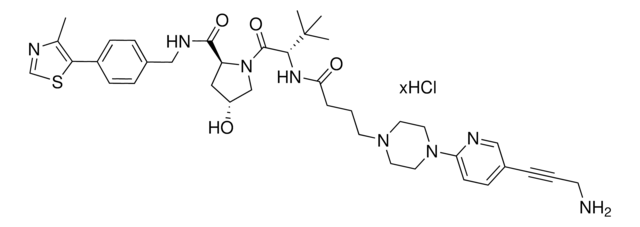
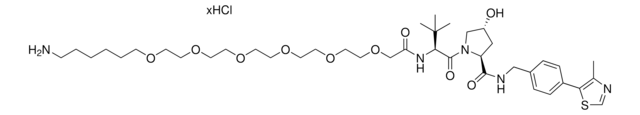
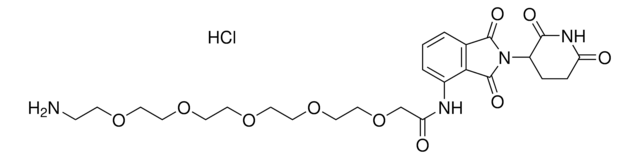
(S,R,S)-AHPC-PEG2-NH2 hydrochloride
≥95%
Sinônimo(s):
(2S,4R)-1-((S)-2-(2-(2-(2-Aminoethoxy)ethoxy)acetamido)-3,3-dimethylbutanoyl)-4-hydroxy-N-(4-(4-methylthiazol-5-yl)benzyl)pyrrolidine-2-carboxamide hydrochloride, Crosslinker−E3 Ligase ligand conjugate, Protein degrader building block for PROTAC® research, Template for synthesis of targeted protein degrader, VH032 conjugate
About This Item
Produtos recomendados
ligand
VH032
Nível de qualidade
Ensaio
≥95%
forma
powder or crystals
adequação da reação
reactivity: carboxyl reactive
reagent type: ligand-linker conjugate
grupo funcional
amine
temperatura de armazenamento
2-8°C
cadeia de caracteres SMILES
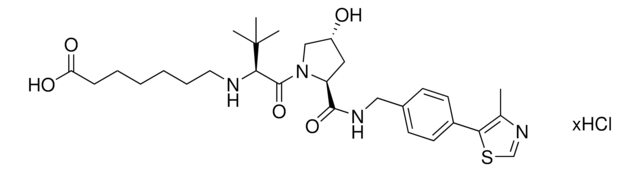
O=C(NCC1=CC=C(C2=C(C)N=CS2)C=C1)[C@H](C[C@@H](O)C3)N3C([C@H](C(C)(C)C)NC(COCCOCCN)=O)=O.Cl
Aplicação
Automate your VHL-PEG based PROTACs with Synple Automated Synthesis Platform (SYNPLE-SC002)
Outras notas
Portal: Building PROTAC® Degraders for Targeted Protein Degradation
Impact of Target Warhead and Linkage Vector on Inducing Protein Degradation: Comparison of Bromodomain and Extra-Terminal (BET) Degraders Derived from Triazolodiazepine (JQ1) and Tetrahydroquinoline (I-BET726) BET Inhibitor Scaffolds
Degradation of the BAF Complex Factor BRD9 by Heterobifunctional Ligands
Targeted Protein Degradation by Small Molecules
Impact of linker length on the activity of PROTACs
Informações legais
produto relacionado
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Certificados de análise (COA)
Busque Certificados de análise (COA) digitando o Número do Lote do produto. Os números de lote e remessa podem ser encontrados no rótulo de um produto após a palavra “Lot” ou “Batch”.
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Artigos
Partial PROTACs are a collection of crosslinker-E3 ligand conjugates with a pendant functional group for covalent linkage to a target ligand.
Partial PROTACs are a collection of crosslinker-E3 ligand conjugates with a pendant functional group for covalent linkage to a target ligand.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica