901558
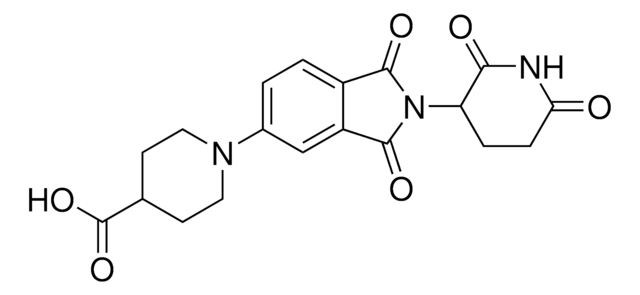
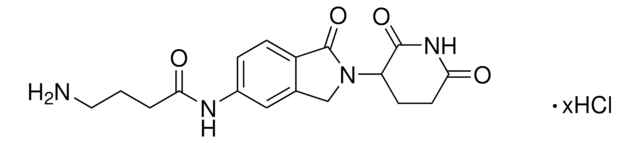
Lenalidomide
≥95%
Sinônimo(s):
1-Oxo-4-amino-2-(2,6-dioxopiperidin-3-yl)isoindole, 3-(4-Amino-1,3-dihydro-1-oxo-2H-isoindol-2-yl)-2,6-piperidinedione, 3-(4-Amino-1-oxoisoindolin-2-yl)piperidine-2,6-dione, E3 Ligase ligand, Ligand for PROTAC® research
About This Item
Produtos recomendados
ligand
lenalidomide
Ensaio
≥95%
forma
powder
adequação da reação
reagent type: ligand
pf
265-268 °C
temperatura de armazenamento
2-8°C
cadeia de caracteres SMILES
O=C1N(C2CCC(NC2=O)=O)CC3=C1C=CC=C3N
InChI
1S/C13H13N3O3/c14-9-3-1-2-7-8(9)6-16(13(7)19)10-4-5-11(17)15-12(10)18/h1-3,10H,4-6,14H2,(H,15,17,18)
chave InChI
GOTYRUGSSMKFNF-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Aplicação
.
Outras notas
Portal: Building PROTAC® Degraders for Targeted Protein Degradation
Structure of the human Cereblon–DDB1–lenalidomide complex reveals basis for responsiveness to thalidomide analogs
Lenalidomide causes selective degradation of IKZF1 and IKZF3 in multiple myeloma cells
The myeloma drug lenalidomide promotes the cereblon-dependent destruction of Ikaros proteins
Informações legais
produto relacionado
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Repr. 1B - STOT RE 2
Órgãos-alvo
Blood
Código de classe de armazenamento
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Artigos
Partial PROTACs are a collection of crosslinker-E3 ligand conjugates with a pendant functional group for covalent linkage to a target ligand.
Partial PROTACs are a collection of crosslinker-E3 ligand conjugates with a pendant functional group for covalent linkage to a target ligand.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica