Key Documents
905356
Rh2(R-PTAD)4
Synonim(y):
Tetrakis[(R)-(-)-(1-adamantyl)-(N-phthalimido)acetato]dirhodium(II)
About This Item
Polecane produkty
Postać
powder or crystals
mp
>300 °C
InChI
1S/4C20H21NO4.2Rh/c4*22-17-14-3-1-2-4-15(14)18(23)21(17)16(19(24)25)20-8-11-5-12(9-20)7-13(6-11)10-20;;/h4*1-4,11-13,16H,5-10H2,(H,24,25);;/q;;;;2*+2/p-4/t4*11-,12+,13-,16-,20?;;/m0000../s1
Klucz InChI
SGEDWOHAUXKUGM-VMLWFVNWSA-J
Zastosowanie
Inne uwagi
Asymmetric Synthesis of Tropanes by Rhodium-Catalyzed [4 + 3] Cycloaddition
Asymmetric [4 + 3] Cycloadditions between Vinylcarbenoids and Dienes: Application to the Total Synthesis of the Natural Product (−)-5-epi-Vibsanin E
produkt powiązany
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Wybierz jedną z najnowszych wersji:
Certyfikaty analizy (CoA)
Przepraszamy, ale COA dla tego produktu nie jest aktualnie dostępny online.
Proszę o kontakt, jeśli potrzebna jest pomoc Obsługa Klienta
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej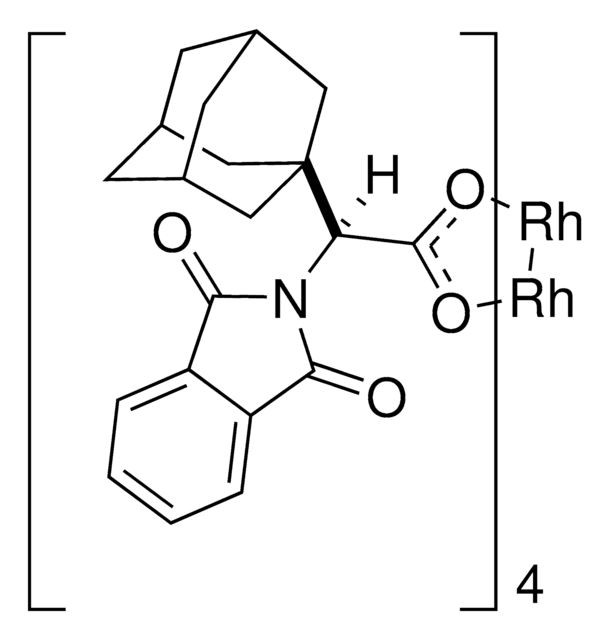

![Bis[rhodium(α,α,α′,α′-tetramethyl-1,3-benzenedipropionic acid)] 95%](/deepweb/assets/sigmaaldrich/product/structures/102/178/d1171a49-0358-406b-8b32-04324dbf9c02/640/d1171a49-0358-406b-8b32-04324dbf9c02.png)