Wszystkie zdjęcia(1)
Key Documents
About This Item
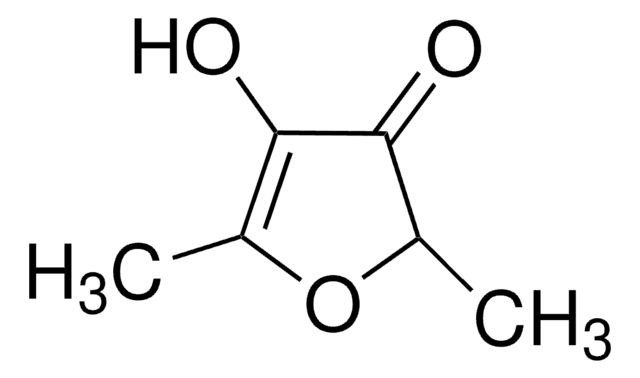
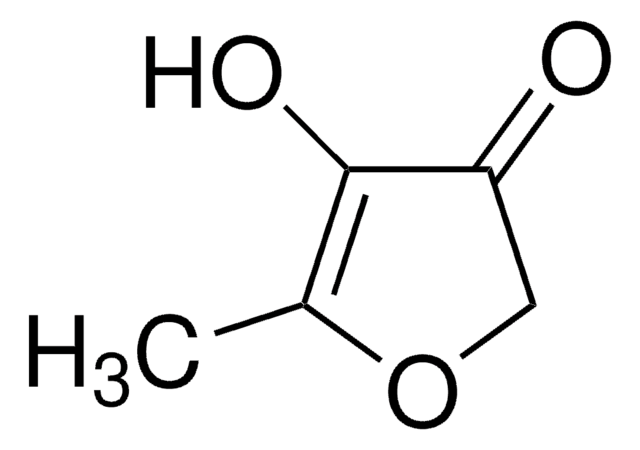
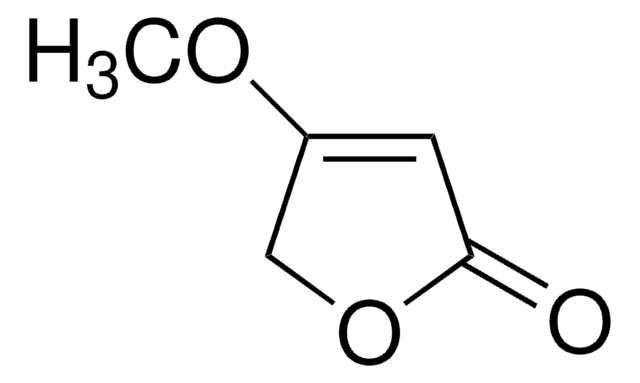
Wzór liniowy:
C4OH2OCH3OH
Numer CAS:
Masa cząsteczkowa:
114.10
Numer WE:
Numer MDL:
Kod UNSPSC:
12352100
Identyfikator substancji w PubChem:
NACRES:
NA.22
Polecane produkty
Próba
97%
mp
129-133 °C (lit.)
ciąg SMILES
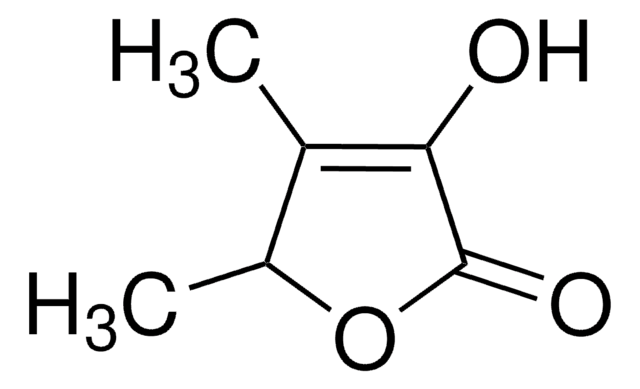
CC1=C(O)C(=O)CO1
InChI
1S/C5H6O3/c1-3-5(7)4(6)2-8-3/h7H,2H2,1H3
Klucz InChI
DLVYTANECMRFGX-UHFFFAOYSA-N
Szukasz podobnych produktów? Odwiedź Przewodnik dotyczący porównywania produktów
Zastosowanie
4-Hydroxy-5-methyl-3-furanone may be used in oxidoreductase assay for the evaluation of oxidoreductase activity.
This page may contain text that has been machine translated.
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Środki ochrony indywidualnej
Eyeshields, Gloves, type N95 (US)
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Highly effective inhibition of biofilm formation by the first metagenome-derived AI-2 quenching enzyme.
Weiland-Brauer N, et al.
Frontiers in Microbiology, 7 (2016)
Reinvestigation of the reaction between 2-furancarboxaldehyde and 4-hydroxy-5-methyl-3(2H)-furanone.
A Ravagli et al.
Journal of agricultural and food chemistry, 47(12), 4962-4969 (1999-12-22)
The reaction between 2-furancarboxaldehyde and 4-hydroxy-5-methyl-3(2H)-furanone was reinvestigated as a part of a systematic study on low molecular weight colored compounds from the Maillard reaction. In acetic acid/piperidine, besides 2-(2-furanylmethylene)-4-hydroxy-5-methyl-3(2H)-furanone (1) and 5-[2-(2-furanyl)ethenyl]-2-(2-furanylmethylene)-4-hydroxy-5-methyl -3( 2H)-furanone (2), four novel compounds, 15a
Tobias Hauck et al.
Journal of agricultural and food chemistry, 51(5), 1410-1414 (2003-02-20)
Formation of the flavor compound and precursor 4-hydroxy-5-methyl-3[2H]-furanone (HMF, norfuraneol) was demonstrated in cytosolic protein extracts obtained from Zygosaccharomyces rouxii after incubation with a number of carbohydrate phosphates. 4-Hydroxy-5-methyl-3[2H]-furanone was produced from d-fructose-1,6-diphosphate, d-fructose-6-phosphate, d-glucose-6-phosphate, 6-phosphogluconate, d-ribose-5-phosphate, and d-ribulose-1,5-diphosphate. Enzyme
F B Whitfield et al.
Journal of agricultural and food chemistry, 47(4), 1626-1634 (1999-11-24)
Reaction of 4-hydroxy-5-methyl-3(2H)-furanone (HMF) with cysteine or hydrogen sulfide at pH 4.5 for 60 min at 140 degrees C produced complex mixtures of volatile compounds, the majority of which contained sulfur. Sixty-nine compounds were identified, some tentatively, by GC/MS. These
H J Kim et al.
Advances in experimental medicine and biology, 434, 91-99 (1998-05-23)
The chemometric principle was used to derive a guideline for obtaining a simple "yes or no" answer about the sterility of food particulates heated at aseptic processing temperatures. A quadratic temperature pulse model was used to estimate bacterial destruction from
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej