140732
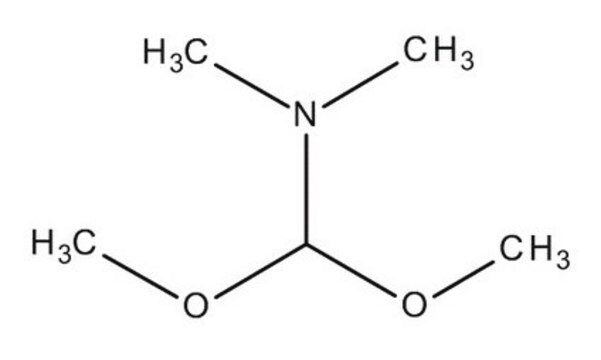
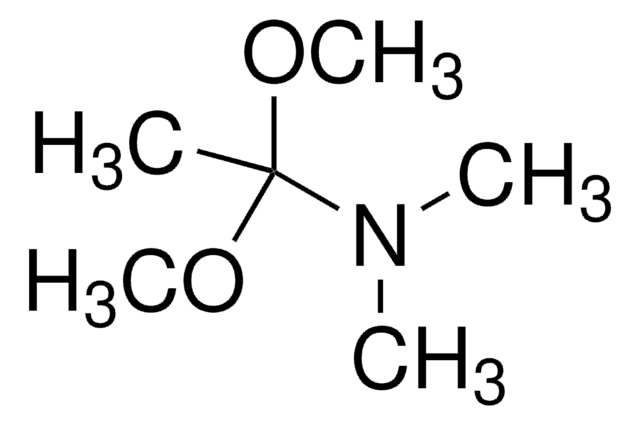
N,N-Dimethylformamide dimethyl acetal
technical grade, 94%
Sinonimo/i:
1,1-Dimethoxy-N,N-dimethylmethylamine, 1,1-Dimethoxytrimethylamine
About This Item
Prodotti consigliati
Grado
technical grade
Livello qualitativo
Saggio
94%
Forma fisica
liquid
Impiego in reazioni chimiche
reaction type: C-C Bond Formation
Indice di rifrazione
n20/D 1.396 (lit.)
P. eboll.
102-103 °C/720 mmHg (lit.)
Densità
0.897 g/mL at 25 °C (lit.)
Gruppo funzionale
amine
ether
Stringa SMILE
COC(OC)N(C)C
InChI
1S/C5H13NO2/c1-6(2)5(7-3)8-4/h5H,1-4H3
ZSXGLVDWWRXATF-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Applicazioni
- An organocatalyst for the synthesis of 1,2-disubstituted-3,4-dihydronaphthalenes by the reaction of vinylarenes and electron-deficient alkynes.
- A building block for the preparation of heterocyclic compounds such as enamines and amidines via formylation of methylene and amino groups.
- A reagent in the synthesis of 1,4-diaryl-piperazine-2,5-diones by reacting with chloroacetamides.
- A reagent along with enamine and alkyne for the preparation of 2,3,4,5-tetrasubstituted pyridine derivatives by three-component coupling reaction in the presence of Me3SiCl.
Avvertenze
Danger
Indicazioni di pericolo
Classi di pericolo
Acute Tox. 4 Inhalation - Eye Dam. 1 - Flam. Liq. 2 - Skin Sens. 1
Codice della classe di stoccaggio
3 - Flammable liquids
Classe di pericolosità dell'acqua (WGK)
WGK 1
Punto d’infiammabilità (°F)
42.8 °F
Punto d’infiammabilità (°C)
6 °C
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.