343412
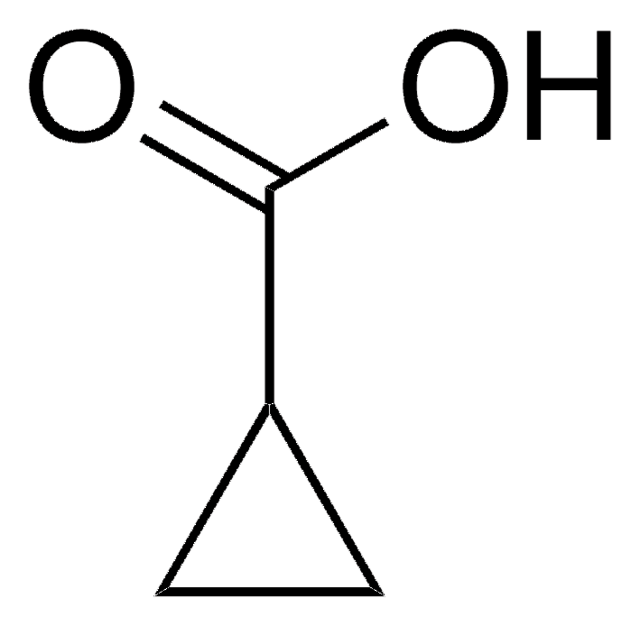
Cyclopropane-1,1-dicarboxylic acid
97%
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula linear:
C3H4(CO2H)2
Número CAS:
Peso molecular:
130.10
Beilstein:
1864823
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Ensaio
97%
pf
134-136 °C (lit.)
solubilidade
methanol: soluble 1 g/10 mL, clear, colorless
grupo funcional
carboxylic acid
cadeia de caracteres SMILES
OC(=O)C1(CC1)C(O)=O
InChI
1S/C5H6O4/c6-3(7)5(1-2-5)4(8)9/h1-2H2,(H,6,7)(H,8,9)
chave InChI
FDKLLWKMYAMLIF-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
Cyclopropane-1,1-dicarboxylic acid is a dicarboxylic acid. Cyclopropane-1,1-dicarboxylic acid, an inhibitor of 1-aminocyclopropane-1-carboxylic acid oxidase, was quantitated in Lycopersicum esculentum by HPLC-electrospray tandem mass spectrometry. Crystal and molecular structure of cyclopropane-1,1-dicarboxylic acid has been reported.
Aplicação
Cyclopropane-1,1-dicarboxylic acid was used in the preparation of new heterocyclic derivatives of cyclopropane dicarboxylic acid containing thiadiazole and 1,2,4-triazole moieties. It was also used to prepare spiro-cyclopropyl metallocycles.
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Skin Corr. 1B
Código de classe de armazenamento
8A - Combustible corrosive hazardous materials
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
The crystal and molecular structure of cyclopropane-1, 1-dicarboxylic acid.
Meester MAM, et al.
Acta Crystallographica Section B, Structural Science, 27(3), 630-634 (1971)
Tetrahedron, 45, 1219-1219 (1989)
Paolo Ingallinella et al.
Biochemistry, 41(17), 5483-5492 (2002-04-24)
Serine proteases are the most studied class of proteolytic enzymes and a primary target for drug discovery. Despite the large number of inhibitors developed so far, very few make contact with the prime site of the enzyme, which constitutes an
Nora A Foroud et al.
Phytopathology, 109(5), 796-803 (2018-12-13)
Plant signaling hormones such as ethylene have been shown to affect the host response to various pathogens. Often, the resistance responses to necrotrophic fungi are mediated through synergistic interactions of ethylene (ET) with the jasmonate signaling pathway. On the other
Yan-Biao Kang et al.
Organic & biomolecular chemistry, 4(2), 299-301 (2006-01-05)
A tandem ring-opening-cyclization reaction of cyclopropanes with imines in the presence of 5 mol% of scandium triflate was developed for the highly diastereoselective synthesis of multi-substituted pyrrolidines.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica