Kluczowe dokumenty
52650
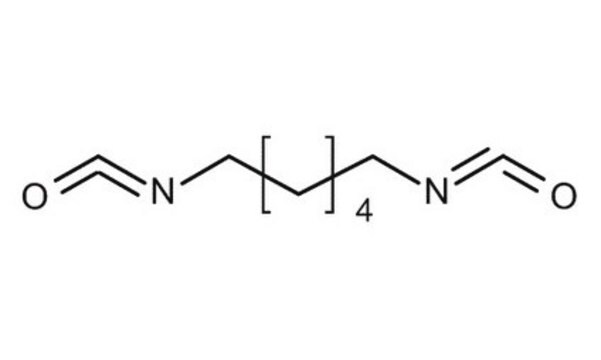
Hexamethylene diisocyanate
purum, ≥98.0% (GC)
Synonim(y):
1,6-Diisocyanatohexane
About This Item
Polecane produkty
klasa czystości
purum
Poziom jakości
Próba
≥98.0% (GC)
współczynnik refrakcji
n20/D 1.453
bp
82-85 °C/0.1 mmHg
gęstość
1.047 g/mL at 20 °C (lit.)
ciąg SMILES
O=C=NCCCCCCN=C=O
InChI
1S/C8H12N2O2/c11-7-9-5-3-1-2-4-6-10-8-12/h1-6H2
Klucz InChI
RRAMGCGOFNQTLD-UHFFFAOYSA-N
Szukasz podobnych produktów? Odwiedź Przewodnik dotyczący porównywania produktów
Powiązane kategorie
Opis ogólny
Zastosowanie
- A crosslinker to crosslink the polyurethane chains in the triblock copolymer gate dielectric, which is then deposited on the substrate to fabricate low-voltage organic thin-film transistors.
- A precursor in the preparation of electroactive shape memory polyurethane/graphene nanocomposites. These materials are usually used as actuators, sensors, artificial muscles, smart devices, and microswitches.
- A crosslinker in conjunction with Pluronic F127, a nonionic surfactant, to synthesize a poly(lactic acid) (PLA)-based hydrogel for biomedical applications.
Hasło ostrzegawcze
Danger
Zwroty wskazujące rodzaj zagrożenia
Zwroty wskazujące środki ostrożności
Klasyfikacja zagrożeń
Acute Tox. 1 Inhalation - Acute Tox. 4 Oral - Eye Dam. 1 - Resp. Sens. 1 - Skin Corr. 1C - Skin Sens. 1 - STOT SE 3
Organy docelowe
Respiratory system
Kod klasy składowania
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
Klasa zagrożenia wodnego (WGK)
WGK 1
Temperatura zapłonu (°F)
266.0 °F - Pensky-Martens closed cup
Temperatura zapłonu (°C)
130 °C - Pensky-Martens closed cup
Środki ochrony indywidualnej
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Wykazy regulacyjne
Wykazy regulacyjne dotyczą głównie produktów chemicznych. Można w nich podawać ograniczoną liczbę informacji na temat produktów niechemicznych. Brak wpisu oznacza, że żaden ze składników nie znajduje się w wykazie. Użytkownik odpowiada za zagwarantowanie bezpiecznego i zgodnego z prawem stosowania produktu.
EU REACH Annex XVII (Restriction List)
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Protokoły
HPLC Analysis of Isocyanates on Titan™ C18
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej