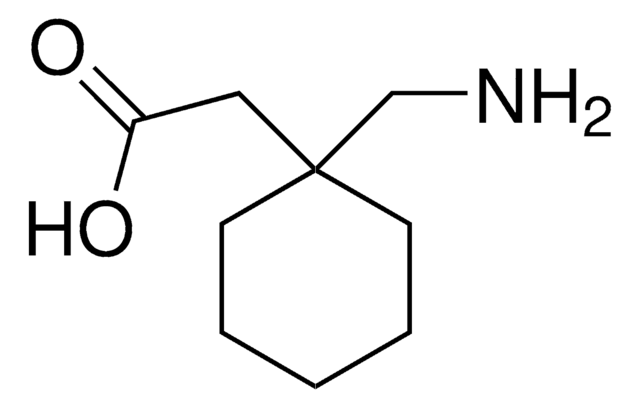
おすすめの製品
グレード
certified reference material
pharmaceutical secondary standard
品質水準
認証
traceable to BP 28
traceable to Ph. Eur. B0200000
traceable to USP 1048200
APIファミリー
baclofen
CofA
current certificate can be downloaded
包装
pkg of 1 g
テクニック
HPLC: suitable
gas chromatography (GC): suitable
アプリケーション
pharmaceutical (small molecule)
フォーマット
neat
保管温度
2-30°C
SMILES記法
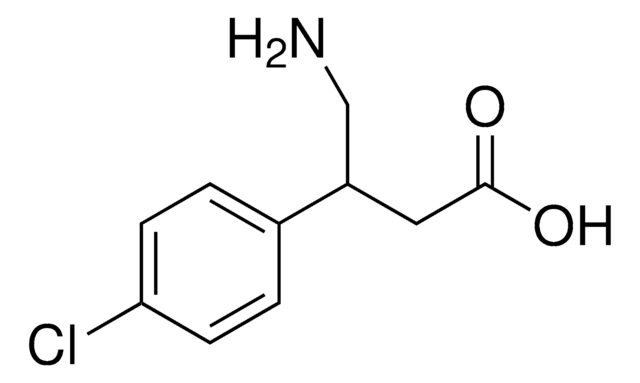
ClC1=CC=C(C(CN)CC(O)=O)C=C1
InChI
1S/C10H12ClNO2/c11-9-3-1-7(2-4-9)8(6-12)5-10(13)14/h1-4,8H,5-6,12H2,(H,13,14)
InChI Key
KPYSYYIEGFHWSV-UHFFFAOYSA-N
遺伝子情報
human ... GABBR1(2550) , GABBR2(9568)
類似した製品をお探しですか? 訪問 製品比較ガイド
詳細
Baclofen is a structural analog of γ-aminobutyric acid (GABA) and an antispastic agent commonly used for the treatment of spasticity as a result of spinal cord or brain injury.
アプリケーション
生物化学的/生理学的作用
アナリシスノート
その他情報
脚注
おすすめ製品
関連製品
シグナルワード
Danger
危険有害性情報
危険有害性の分類
Acute Tox. 3 Oral
保管分類コード
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
PHR1682-1G:
PHR1682-1G-PW:
最新バージョンのいずれかを選択してください:
この製品を見ている人はこちらもチェック
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)