About This Item
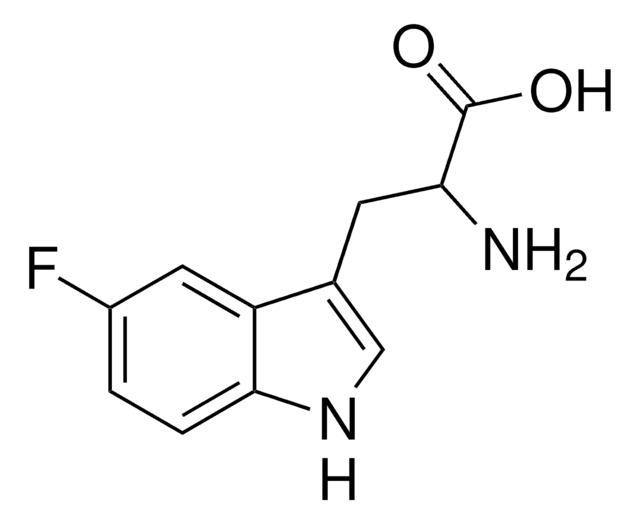
Fórmula empírica (Notação de Hill):
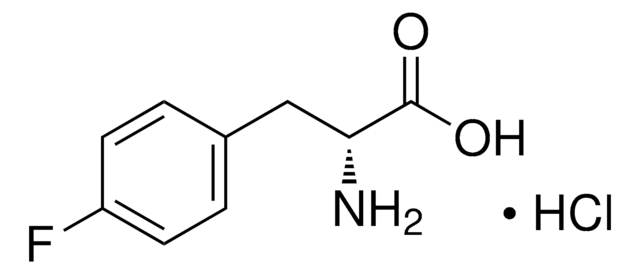
C11H11FN2O2
Número CAS:
Peso molecular:
222.22
Beilstein:
5052680
Número MDL:
Código UNSPSC:
12352202
eCl@ss:
32160406
ID de substância PubChem:
NACRES:
NA.77
Produtos recomendados
Ensaio
≥98.0% (HPLC)
Formulário
powder
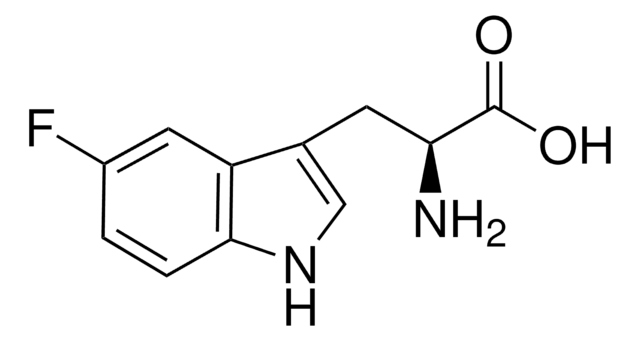
pureza óptica
enantiomeric ratio: ≥99.5:0.5 (HPLC)
pf
270-280 °C
temperatura de armazenamento
2-8°C
cadeia de caracteres SMILES
N[C@@H](Cc1c[nH]c2ccc(F)cc12)C(O)=O
InChI
1S/C11H11FN2O2/c12-7-1-2-10-8(4-7)6(5-14-10)3-9(13)11(15)16/h1-2,4-5,9,14H,3,13H2,(H,15,16)/t9-/m0/s1
chave InChI
INPQIVHQSQUEAJ-VIFPVBQESA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Aplicação
Exogenous 5-fluoro-Trp is incorporated into proteins in normal protein synthesis. Since 19F is a useful reporter group, this provides a method for studying enzyme mechanisms by NMR.
Ações bioquímicas/fisiológicas
5-Fluoro-Trp is nonspecifically cytotoxic. It is believed this is due to malfunctioning enzymes that have had replacements of Trp residues by 5-fluoro-Trp. However, at least one case is known where 5-fluoro-Trp substitution leads to significantly greater catalytic activity.
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
G S Rule et al.
Biochemistry, 26(2), 549-556 (1987-01-27)
In this study we demonstrate the potential of combining fluorine-19 nuclear magnetic resonance (NMR) spectroscopy with molecular genetics. We are using the membrane-bound enzyme D-lactate dehydrogenase of Escherichia coli as a model system to characterize interactions between proteins and lipids.
Dereje Abate Negatu et al.
mBio, 10(2) (2019-03-28)
Indole propionic acid (IPA), produced by the gut microbiota, is active against Mycobacterium tuberculosisin vitro and in vivo However, its mechanism of action is unknown. IPA is the deamination product of tryptophan (Trp) and thus a close structural analog of
E W Miles et al.
Biochemistry, 25(15), 4240-4249 (1986-07-29)
We are exploring the active site and the mechanism of the pyridoxal phosphate dependent reactions of the bacterial tryptophan synthase alpha 2 beta 2 complex by use of substrate analogues and of reaction intermediate analogues. Fluorine-19 nuclear magnetic resonance studies
Warintra Pitsawong et al.
eLife, 7 (2018-06-15)
Protein kinases are major drug targets, but the development of highly-selective inhibitors has been challenging due to the similarity of their active sites. The observation of distinct structural states of the fully-conserved Asp-Phe-Gly (DFG) loop has put the concept of
S Rozovsky et al.
Journal of molecular biology, 310(1), 271-280 (2001-06-23)
Product release is partially rate determining in the isomerization reaction catalyzed by Triosephosphate Isomerase, the conversion of dihydroxyacetone phosphate to D-glyceraldehyde 3-phosphate, probably because an active-site loop movement is necessary to free the product from confinement in the active-site. The
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica