PHR1847
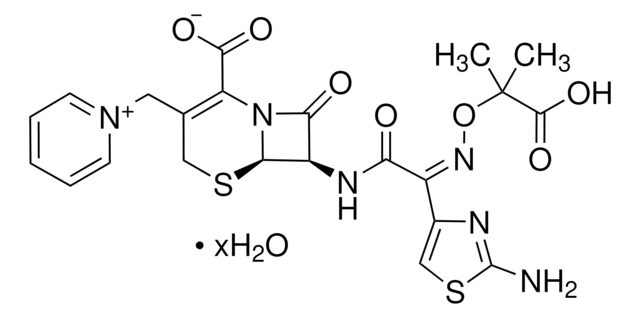
Ceftazidime
Pharmaceutical Secondary Standard; Certified Reference Material
Sinônimo(s):
Ceftazidime pentahydrate
About This Item
Produtos recomendados
grau
certified reference material
pharmaceutical secondary standard
Nível de qualidade
Agency
traceable to Ph. Eur. C0690500
traceable to USP 1098130
família API
ceftazidime
Formulário
solid
Certificado de análise (CofA)
current certificate can be downloaded
embalagem
pkg of 500 mg
técnica(s)
HPLC: suitable
gas chromatography (GC): suitable
aplicação(ões)
pharmaceutical (small molecule)
temperatura de armazenamento
-10 to -25°C
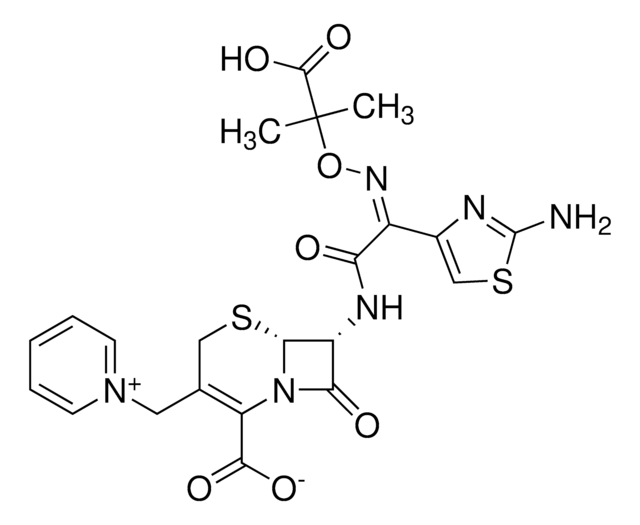
cadeia de caracteres SMILES
[s]1c(nc(c1)\C(=N\OC(C)(C)C(=O)[O-])\C(=O)N[C@H]2[C@H]3SCC(=C(N3C2=O)C(=O)O)C[n+]4ccccc4)N.O.O.O.O.O
InChI
1S/C22H22N6O7S2.5H2O/c1-22(2,20(33)34)35-26-13(12-10-37-21(23)24-12)16(29)25-14-17(30)28-15(19(31)32)11(9-36-18(14)28)8-27-6-4-3-5-7-27;;;;;/h3-7,10,14,18H,8-9H2,1-2H3,(H4-,23,24,25,29,31,32,33,34);5*1H2/b26-13-;;;;;/t14-,18-;;;;;/m1...../s1
chave InChI
NMVPEQXCMGEDNH-TZVUEUGBSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
Aplicação
Nota de análise
Outras notas
Nota de rodapé
produto relacionado
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Resp. Sens. 1 - Skin Sens. 1
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Certificados de análise (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documentos section.
Se precisar de ajuda, entre em contato Atendimento ao cliente
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica