M1650000
Metil 4-hidroxibenzoato
European Pharmacopoeia (EP) Reference Standard
Sinônimo(s):
Éster metílico de ácido p-hidroxibenzoico, Metilparabeno, NIPAGIN
About This Item
Produtos recomendados
grau
pharmaceutical primary standard
família API
parabens
fabricante/nome comercial
EDQM
pf
125-128 °C (lit.)
aplicação(ões)
pharmaceutical (small molecule)
Formato
neat
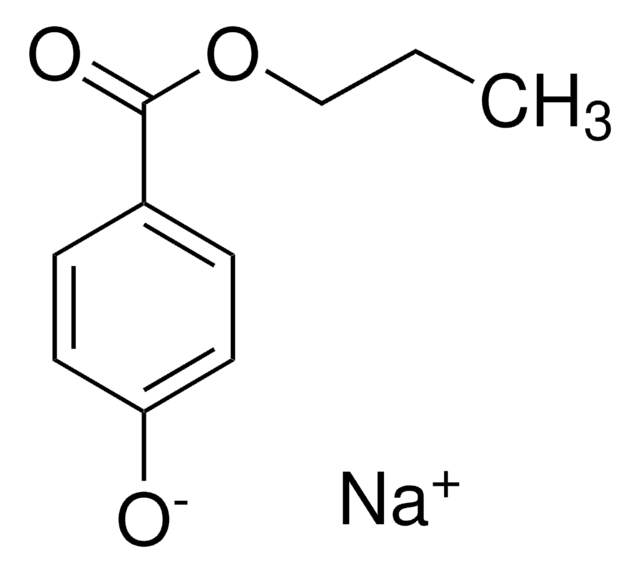
cadeia de caracteres SMILES
COC(=O)c1ccc(O)cc1
InChI
1S/C8H8O3/c1-11-8(10)6-2-4-7(9)5-3-6/h2-5,9H,1H3
chave InChI
LXCFILQKKLGQFO-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Descrição geral
For further information and support please go to the website of the issuing Pharmacopoeia.
Aplicação
Established for use according to European Pharmacopoeia for the preparation of the below-given solutions:
- Reference solutions (a) and (b) in the identification, testing for related substances, and assay of methyl parahydroxybenzoate and sodium methyl parahydroxybenzoate, according to the monographs 0409 and 1262
- Reference solution (c) for the testing of related substances in nifuroxazide using liquid chromatography (2.2.29), according to the monograph 1999
- Reference solution (b) in the identification of sodium ethyl parahydroxybenzoate using thin-layer chromatography (General text 2.2.27), according to the monograph 2134
Embalagem
Outras notas
produto relacionado
Frases de perigo
Declarações de precaução
Classificações de perigo
Aquatic Chronic 2
Código de classe de armazenamento
13 - Non Combustible Solids
Classe de risco de água (WGK)
WGK 1
Ponto de fulgor (°F)
334.4 °F
Ponto de fulgor (°C)
168 °C
Escolha uma das versões mais recentes:
Certificados de análise (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documentos section.
Se precisar de ajuda, entre em contato Atendimento ao cliente
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica