T71501
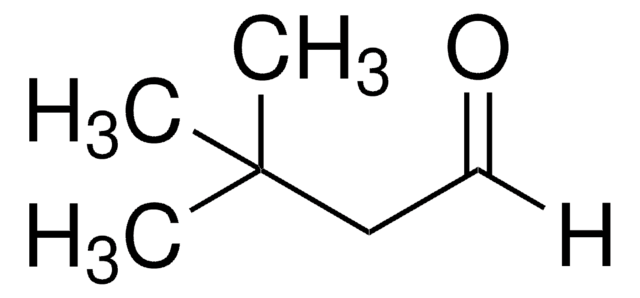
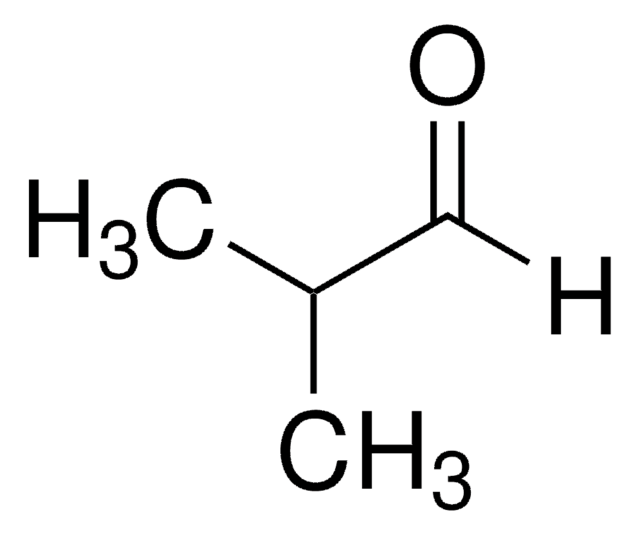
Trimethylacetaldehyde
96%
Sinônimo(s):
Pivalaldehyde, Trimethylacetaldehyde
About This Item
Produtos recomendados
Nível de qualidade
Ensaio
96%
índice de refração
n20/D 1.378 (lit.)
p.e.
74 °C/730 mmHg (lit.)
pf
6 °C (lit.)
densidade
0.793 g/mL at 25 °C (lit.)
temperatura de armazenamento
2-8°C
cadeia de caracteres SMILES
[H]C(=O)C(C)(C)C
InChI
1S/C5H10O/c1-5(2,3)4-6/h4H,1-3H3
chave InChI
FJJYHTVHBVXEEQ-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Aplicação
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Eye Irrit. 2 - Flam. Liq. 2 - Skin Irrit. 2 - STOT SE 3
Órgãos-alvo
Respiratory system
Código de classe de armazenamento
3 - Flammable liquids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
3.2 °F - closed cup
Ponto de fulgor (°C)
-16 °C - closed cup
Equipamento de proteção individual
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Global Trade Item Number
| SKU | GTIN |
|---|---|
| T71501-100ML | 4061836696856 |
| T71501-500ML | |
| T71501-5ML | 4061837372322 |
| T71501-25G | |
| T71501-25ML | 4061837372315 |
| T71501-5G |
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica