T9003
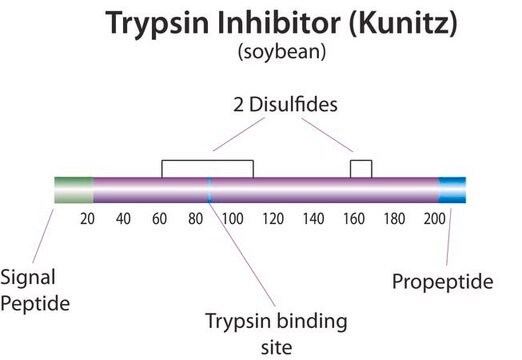
Trypsin inhibitor
lyophilized powder
Synonym(s):
SBTI
About This Item
Recommended Products
Product Name
Trypsin inhibitor from Glycine max (soybean), lyophilized powder
biological source
Glycine max (soybean)
Quality Level
form
lyophilized powder
mol wt
20.1 kDa
solubility
H2O: >10 mg/mL
phosphate buffer: >10 mg/mL (hazy above 10 mg/ml)
soluble
application(s)
diagnostic assay manufacturing
storage temp.
2-8°C
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
Biochem/physiol Actions
Components
Unit Definition
Preparation Note
Solutions can retain activity when stored short-term at 2-8° C. Solutions are stable in frozen aliquots at -20°C.
Analysis Note
signalword
Danger
hcodes
Hazard Classifications
Resp. Sens. 1 - Skin Sens. 1
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
This technical article described the Enzymatic Assay of Trypsin Inhibitor.
Related Content
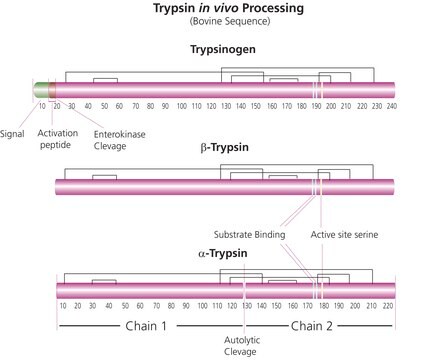
Trypsin is an enzyme in the serine protease class that consists of a polypeptide chain of 223 amino acid residues. Multiple sources, grades and formulations of trypsin specifically designed for research applications are available.
Chromatograms
application for HPLCOur team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service