03449
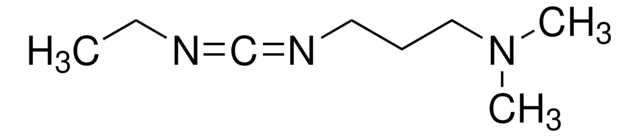
N-(3-Dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride
≥99.0% (AT)
Synonym(s):
N-Ethyl-N′-(3-dimethylaminopropyl)carbodiimide hydrochloride, EDAC, EDC, EDC hydrochloride, WSC hydrochloride
About This Item
Recommended Products
Quality Level
assay
≥99.0% (AT)
form
powder
technique(s)
bioconjugation: suitable
mp
110-115 °C (lit.)
112-116 °C
solubility
H2O: soluble 0.2 g/L
storage temp.
−20°C
SMILES string
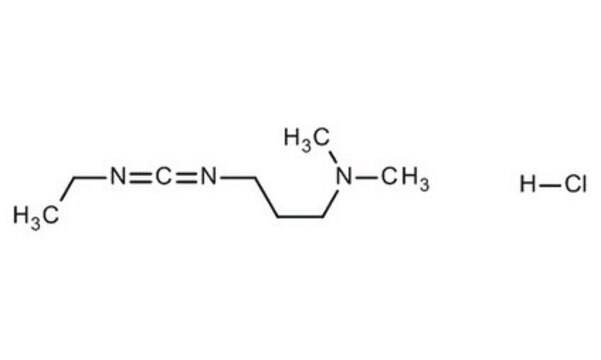
Cl.CCN=C=NCCCN(C)C
InChI
1S/C8H17N3.ClH/c1-4-9-8-10-6-5-7-11(2)3;/h4-7H2,1-3H3;1H
InChI key
FPQQSJJWHUJYPU-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Furthermore, EDAC HCl serves as a biomolecule bridge, acting as a crosslinker that connects amine-reactive NHS-esters of biomolecules to carboxyl groups. This technique proves invaluable in protein conjugation, enabling the creation of hybrid molecules with novel properties and functions. The underlying mechanism involves EDAC HCl′s reaction with a carboxyl group, forming an unstable intermediate that actively seeks an amine partner. The delicate balance of this reaction underscores the importance of optimizing conditions for efficient conjugation. The assistance of N-hydroxysuccinimide (NHS) further enhances EDAC HCl′s capabilities by stabilizing the intermediate and enabling two-step conjugation procedures. This additional feature provides greater flexibility and control, particularly when dealing with complex biomolecules.
Application
Biochem/physiol Actions
Features and Benefits
Other Notes
Comparable product
signalword
Danger
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Skin Irrit. 2 - Skin Sens. 1 - STOT RE 2 Oral
target_organs
Stomach,large intestine,lymph node
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Carbodiimide-mediated peptide coupling remains to the most frequently used technique.
Amide bonds are ubiquitous in both nature and industrial applications. They are vital to the structure and function of biological macromolecules and polymers. The importance of this functionality has resulted in numerous approaches to its formation, ranging from stoichiometric activation of carboxylic acids to more recent advances in catalytic amide bond formation.
Professor Aran (Claremont University, USA) thoroughly discusses the engineering of graphene based materials through careful functionalization of graphene oxide, a solution processable form of graphene.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service






![1-[3-(Dimethylamino)propyl]-3-ethylcarbodiimide methiodide](/deepweb/assets/sigmaaldrich/product/structures/414/134/4eb9c126-d7f9-4e12-9e3a-95cb077824fd/640/4eb9c126-d7f9-4e12-9e3a-95cb077824fd.png)