DTT-RO
Roche
DTT
crystalline powder, =97% (Ellman′s reagent), Mr 154.3
Synonym(s):
DL-Dithiothreitol, 1,4-dithiothreitol, cleland′s reagent, dithiothreitol, 1,4-, threo-1,4-Dimercapto-2,3-butanediol, Cleland’s reagent, DTT
About This Item
Recommended Products
description
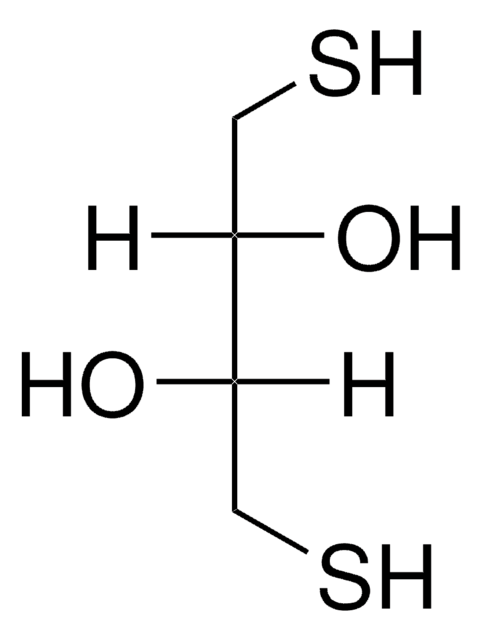
1,4-Dithiothreitol
assay
97% (Ellman′s reagent)
form
crystalline powder
mol wt
Mr 154.3
reaction suitability
reagent type: reductant
packaging
pkg of 10 g (10708984001)
pkg of 2 g (10197777001)
pkg of 25 g (11583786001)
manufacturer/tradename
Roche
pH
5.1 (20 °C)
mp
41-44 °C (lit.)
storage temp.
2-8°C
SMILES string
O[C@H](CS)[C@H](O)CS
InChI
1S/C4H10O2S2/c5-3(1-7)4(6)2-8/h3-8H,1-2H2/t3-,4-/m1/s1
InChI key
VHJLVAABSRFDPM-QWWZWVQMSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- Isolation, purification and characterization of proteins and enzymes
- Measurement of enzyme activities (reactivation of enzymes)
- Determination of disulfide groups in proteins and enzymes
- DNA extraction prior to amplification
Specifications
Sequence
Our DTT-preparation is optically inactive, i.e. it is the D,L-DTT.
Preparation Note
Other Notes
signalword
Danger
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Chronic 3 - Eye Dam. 1 - Skin Irrit. 2
Storage Class
11 - Combustible Solids
wgk_germany
WGK 2
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service