130672
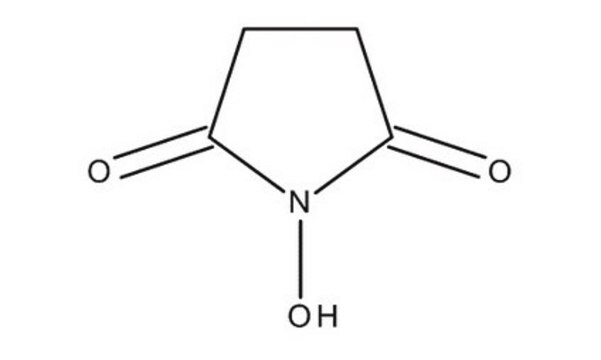
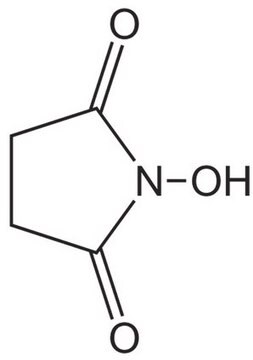
N-Hydroxysuccinimide
98%
Synonym(s):
1-Hydroxy-2,5-pyrrolidinedione, HOSu, NHS
About This Item
Recommended Products
Quality Level
assay
98%
form
solid
mp
95-98 °C (lit.)
functional group
imide
SMILES string
ON1C(=O)CCC1=O
InChI
1S/C4H5NO3/c6-3-1-2-4(7)5(3)8/h8H,1-2H2
InChI key
NQTADLQHYWFPDB-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- N
- -succinimidyl 3-(di-tert-butylfluorosilyl)benzoate
- 4-[2,2-bis[(p-tolylsulfonyl)-methyl] acetyl]benzoic acid-NHS ester
- N-succinimidyl 3-iodobenzoate
It has also been used in a protocol for the surface modification of microchannels of a microfluidic-integrated surface plasmon resonance (SPR) platform for the detection and quantification of bacterial pathogens.
signalword
Danger
hcodes
Hazard Classifications
Eye Dam. 1 - Skin Irrit. 2
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
This article briefly reviews the methods and mechanisms for the formation of molecular monolayers on silicon surfaces, the properties of these monolayers and current perspectives regarding their application in molecular electronic and sensing applications.
Professor Aran (Claremont University, USA) thoroughly discusses the engineering of graphene based materials through careful functionalization of graphene oxide, a solution processable form of graphene.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
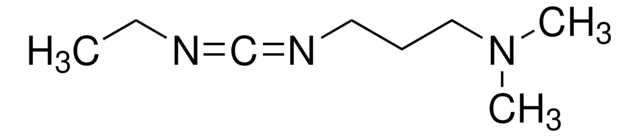




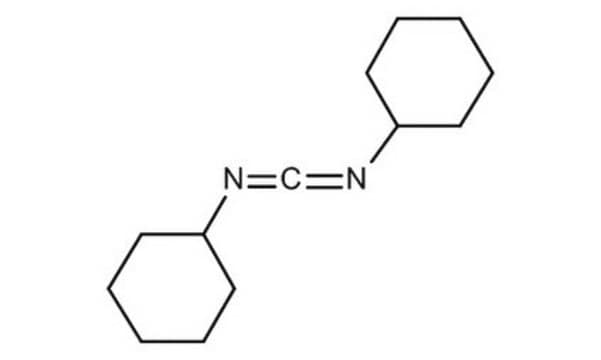

![1-[3-(Dimethylamino)propyl]-3-ethylcarbodiimide methiodide](/deepweb/assets/sigmaaldrich/product/structures/414/134/4eb9c126-d7f9-4e12-9e3a-95cb077824fd/640/4eb9c126-d7f9-4e12-9e3a-95cb077824fd.png)