Key Documents
E7521
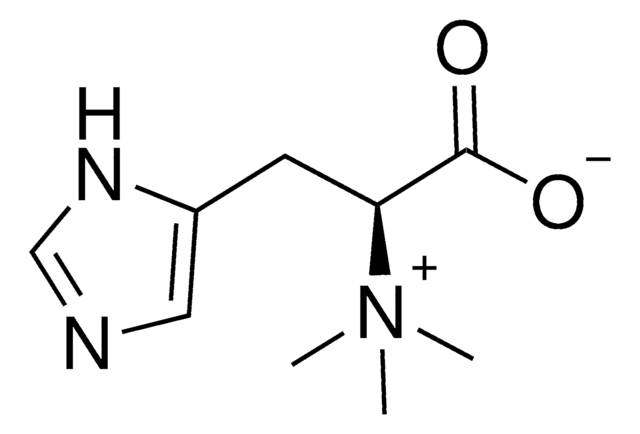
L-(+)-Ergothioneine
Synonim(y):
2-mercaptohistidine trimethyl betaine, Ergothioneine, Sympectothion, Thiasine, Thiolhistidine-betaine, Thioneine, (S)-α-Carboxy-N,N,N-trimethyl-2-mercapto-1H-imidazole-4-ethanaminium inner salt
About This Item
Polecane produkty
pochodzenie biologiczne
fungus (Actinomycetales)
fungus (Ascomycota)
fungus (Basidiomycota)
Poziom jakości
Próba
≥98.0%
Postać
powder
masa cząsteczkowa
229.30
warunki przechowywania
(Keep container tightly closed in a dry and well-ventilated place)
metody
protein quantification: suitable
rozpuszczalność
water: 50 mg/mL, clear, colorless
temp. przechowywania
−20°C
ciąg SMILES
C[N+](C)(C)[C@@H](Cc1c[nH]c(S)n1)C([O-])=O
InChI
1S/C9H15N3O2S/c1-12(2,3)7(8(13)14)4-6-5-10-9(15)11-6/h5,7H,4H2,1-3H3,(H2-,10,11,13,14,15)/t7-/m0/s1
Klucz InChI
SSISHJJTAXXQAX-ZETCQYMHSA-N
Szukasz podobnych produktów? Odwiedź Przewodnik dotyczący porównywania produktów
Opis ogólny
Research area: Apoptosis
Zastosowanie
L-(+)-Ergothioneine has been used:
- as a component of the maturation medium for cumulus-oocyte complexes (COCs) to test protective function on lipid peroxide formation
- as an antioxidant compound to test type 2 diabetes patients
- as a positive control in solute carrier protein 22 A4 (SLC22A4) transport assay
Działania biochem./fizjol.
Opakowanie
Inne uwagi
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Środki ochrony indywidualnej
Eyeshields, Gloves, type N95 (US)
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej