911666
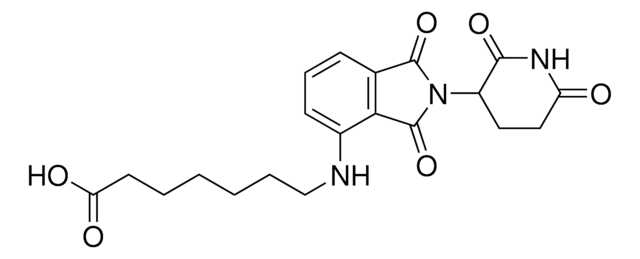
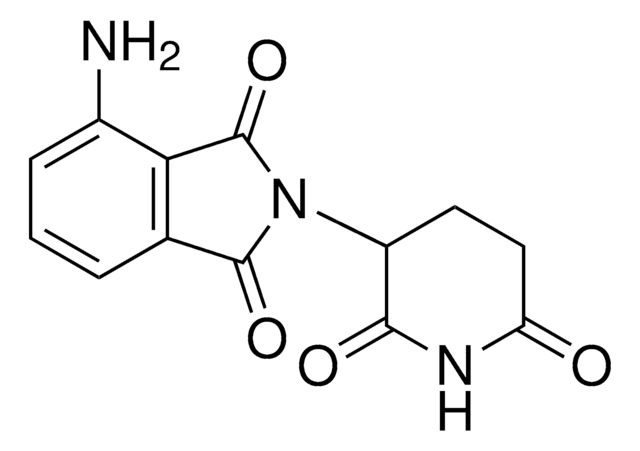
Pomalidomide-C6-NH2 hydrochloride
≥95%
Sinonimo/i:
4-((6-Aminohexyl)amino)-2-(2,6-dioxopiperidin-3-yl)isoindoline-1,3-dione hydrochloride, Crosslinker−E3 ligase ligand conjugate, Pomalidomide conjugate, Protein degrader building block for PROTAC® research, Template for synthesis of targeted protein degrader
About This Item
Prodotti consigliati
ligand
pomalidomide
Saggio
≥95%
Stato
powder
Impiego in reazioni chimiche
reactivity: carboxyl reactive
reagent type: ligand-linker conjugate
Gruppo funzionale
amine
Temperatura di conservazione
2-8°C
Stringa SMILE
O=C(C(CC1)N(C2=O)C(C3=C2C=CC=C3NCCCCCCN)=O)NC1=O.Cl
InChI
1S/C19H24N4O4.ClH/c20-10-3-1-2-4-11-21-13-7-5-6-12-16(13)19(27)23(18(12)26)14-8-9-15(24)22-17(14)25;/h5-7,14,21H,1-4,8-11,20H2,(H,22,24,25);1H
PBGMRXNTLPSDNR-UHFFFAOYSA-N
Categorie correlate
Applicazioni
Altre note
Portal: Building PROTAC® Degraders for Targeted Protein Degradation
Proteolysis Targeting Chimeras for the Selective Degradation of Mcl-1/Bcl-2 Derived from Nonselective Target Binding Ligands
Chemoselective Synthesis of Lenalidomide-Based PROTAC Library Using Alkylation Reaction
Identification of New Small-Molecule Inducers of Estrogen-related Receptor α (ERRα) Degradation
Discovery of MD-224 as a First-in-Class, Highly Potent and Efficacious PROTAC MDM2 Degrader Capable of Achieving Complete and Durable Tumor Regression
Note legali
Prodotti correlati
Avvertenze
Danger
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Repr. 1B
Codice della classe di stoccaggio
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documenti section.
Se ti serve aiuto, non esitare a contattarci Servizio Clienti
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Articoli
Partial PROTACs are a collection of crosslinker-E3 ligand conjugates with a pendant functional group for covalent linkage to a target ligand.
Partial PROTACs are a collection of crosslinker-E3 ligand conjugates with a pendant functional group for covalent linkage to a target ligand.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 911666-50MG |
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.