N6287
Nutlin-3
≥98% (HPLC), powder, Mdm2 antagonist
Sinonimo/i:
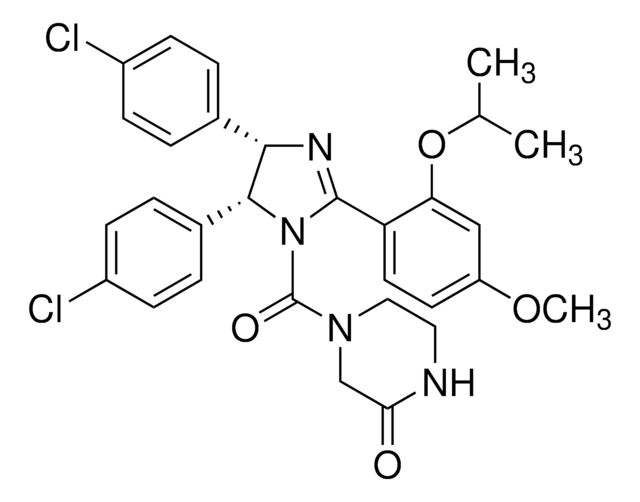
(±)-4-[4,5-Bis(4-chlorophenyl)-2-(2-isopropoxy-4-methoxy-phenyl)-4,5-dihydro-imidazole-1-carbonyl]-piperazin-2-one
About This Item
Prodotti consigliati
Nome del prodotto
Nutlin-3, ≥98% (HPLC), powder
ligand
nutlin-3
Saggio
≥98% (HPLC)
Stato
powder
Impiego in reazioni chimiche
reagent type: ligand
Solubilità
DMSO: 20 mg/mL
H2O: insoluble
Ideatore
Roche
Condizioni di spedizione
wet ice
Temperatura di conservazione
−20°C
Stringa SMILE
O=C(N1CC(NCC1)=O)N2C(C3=CC=C(Cl)C=C3)C(C4=CC=C(Cl)C=C4)N=C2C5=C(OC(C)C)C=C(OC)C=C5
InChI
1S/C30H30Cl2N4O4/c1-18(2)40-25-16-23(39-3)12-13-24(25)29-34-27(19-4-8-21(31)9-5-19)28(20-6-10-22(32)11-7-20)36(29)30(38)35-15-14-33-26(37)17-35/h4-13,16,18,27-28H,14-15,17H2,1-3H3,(H,33,37)/t27-,28+/m1/s1
BDUHCSBCVGXTJM-IZLXSDGUSA-N
Applicazioni
- as a drug to stimulate p53 functions in gene transfer experiment
- to inject worms to verify the prevalent role of translationally controlled tumor protein (TCTP) in posterior amputated E. eugeniae
- as a p53 activator in cyclotherapy studies
- as an mdm2 inhibitor to know its effect on p53 levels, cleaved caspase 3 and Poly (ADP-ribose) polymerase (PARP) cleavage
Azioni biochim/fisiol
Caratteristiche e vantaggi
Altre note
Risultati analitici
Note legali
Prodotti correlati
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.