E7034
EX-527
≥98% (HPLC)
Sinonimo/i:
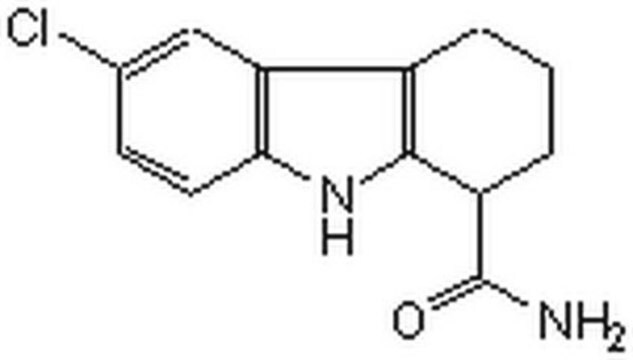
6-Chloro-2,3,4,9-tetrahydro-1H-Carbazole-1-carboxamide
About This Item
Prodotti consigliati
Saggio
≥98% (HPLC)
Stato
powder
Colore
white to beige
Solubilità
DMSO: >20 mg/mL
Temperatura di conservazione
2-8°C
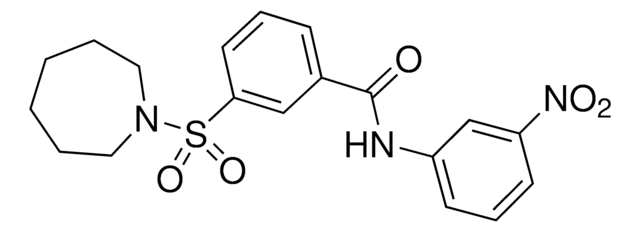
Stringa SMILE
NC(=O)C1CCCc2c1[nH]c3ccc(Cl)cc23
InChI
1S/C13H13ClN2O/c14-7-4-5-11-10(6-7)8-2-1-3-9(13(15)17)12(8)16-11/h4-6,9,16H,1-3H2,(H2,15,17)
FUZYTVDVLBBXDL-UHFFFAOYSA-N
Applicazioni
- in 1% dimethyl sulfoxide, 30%, polyethylene glycol-400 and 1% Tween 80 for treating C57BL/6 N mice to study its effect on intestinal morphological changes and crypt cell apoptosis
- as a an inhibitor of sirtuin 1, in treating human cancer lines MCF-7 (Michigan cancer foundation-7) and HCT116 (colon cancer cell line) incubated in Dulbecco′s modified Eagle′s medium, to study its effect on mitochondrial ATP (adenosine triphosphate) production
- Intracerebroventricularly infused in rat model of epileptogenesis, to access kainic acid–induced status epilepticus stimulated sirtuin 1 activity
Azioni biochim/fisiol
Caratteristiche e vantaggi
Prodotti correlati
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Articoli
Epigenetic modifications are thought to occur through two key interconnected processes—DNA methylation and the covalent modification of histones.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.