G0535
Glycopeptidase A from almonds
buffered aqueous glycerol solution, ≥0.05 unit/mL
Sinônimo(s):
N-Glycosidase A, N-linked-glycopeptide-(N-acetyl-β-D-glucosaminyl)-L-asparagine amidohydrolase, PNGase A
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Número CAS:
Número MDL:
Código UNSPSC:
12352204
NACRES:
NA.32
Produtos recomendados
conjugado
(N-linked)
Nível de qualidade
Formulário
buffered aqueous glycerol solution
concentração
≥0.05 unit/mL
temperatura de armazenamento
−20°C
Descrição geral
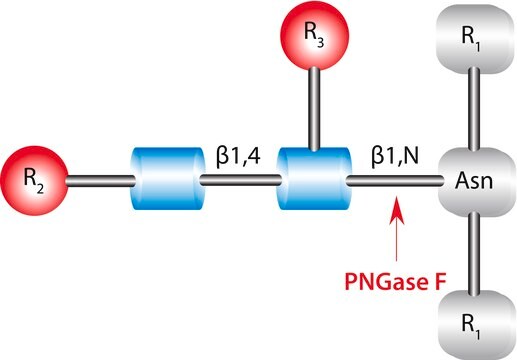
Glycopeptidase found in almonds can be divided into three groups- A, B and C. the optimum pH value and the isoelectric point of glycopeptidase A is 6.0 and 7.7 respectively. It has a preference for glycopeptides with long chains. It is also capable of hydrolyzing intact glycoproteins such as, desialyted human transferrin, ovalbumin etc. These proteins cleave glycoproteins with asialocarbohydrate moieties at their β-aspartyl-glucosylamine linkages.
Aplicação
Glycopeptidase A from almonds is used for deglycosylation. It catalyzes the removal of N-linked oligosaccharide chains and converts Asn residue to Asp.
Ações bioquímicas/fisiológicas
Hydrolyzes an N4-(acetyl-β-D-glycosaminyl)asparagine in which the N-acetyl-D-glucosamine residue may be further glycosylated, yielding a (substituted) N-acetyl-β-D-glucoaminylamine and the peptide containing an aspartic residue.
Definição da unidade
One unit will hydrolyze 1.0 μmole of ovalbumin glycopeptide per min at pH 5.0 at 37°C.
forma física
Solution in 50% glycerol containing 50 mM citrate-phosphate buffer, pH 5.0, and BSA.
Código de classe de armazenamento
10 - Combustible liquids
Classe de risco de água (WGK)
WGK 1
Ponto de fulgor (°F)
No data available
Ponto de fulgor (°C)
No data available
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Karen G Welinder et al.
The Journal of biological chemistry, 284(15), 9764-9769 (2009-02-13)
Proteome data of potato (Solanum tuberosum) tuber juice and of purified potato tuber vacuoles indicated that mature patatins may perhaps lack a C-terminal propeptide. We have confirmed this by complete mass spectrometric sequencing of a number of patatin variants as
Asparagine-linked oligosaccharides in human placenta and umbilical cord as demonstrated by almond glycopeptidase.
N Takahashi et al.
FEBS letters, 146(1), 139-142 (1982-09-06)
T Takahashi et al.
Biochimica et biophysica acta, 657(2), 457-467 (1981-02-13)
The glycopeptidase preparation that has been isolated from almond emulsin and acts on beta-aspartylglycosylamine linkages in glycopeptides was separated into three active fractions by DEAE-cellulose column chromatography. The three discrete species of glycopeptidase (Groups A, B and C) have been
R P Miller et al.
Biochimica et biophysica acta, 954(1), 50-57 (1988-04-28)
The beta-subunit of dog kidney (Na+ + K+)-ATPase is a sialoglycoprotein and contains three potential N-glycosylation sites. In this study, the oligosaccharide chains of purified dog kidney beta-subunit were labeled with tritium by oxidation with sodium periodate or galactose oxidase
Amelie Croset et al.
Journal of biotechnology, 161(3), 336-348 (2012-07-21)
Glycosylation is one of the most common posttranslational modifications of proteins. It has important roles for protein structure, stability and functions. In vivo the glycostructures influence pharmacokinetics and immunogenecity. It is well known that significant differences in glycosylation and glycostructures
Artigos
N-Linked Glycan Strategies; Sigma-Aldrich.com
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica