10810
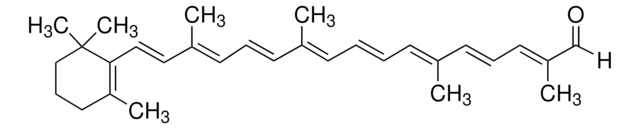
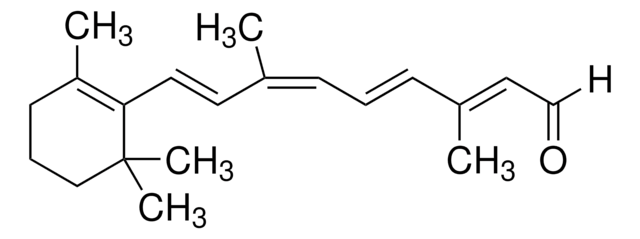
trans-β-Apo-8′-carotenal
≥96.0% (UV)
Sinônimo(s):
Apocarotenal
About This Item
Produtos recomendados
fonte biológica
synthetic
Ensaio
≥96.0% (UV)
Formulário
powder
perda
≤0.5% loss on drying, 20 °C (HV)
pf
137-141 °C
solubilidade
chloroform: 1 mg/mL, clear to very faintly turbid, intense red-orange
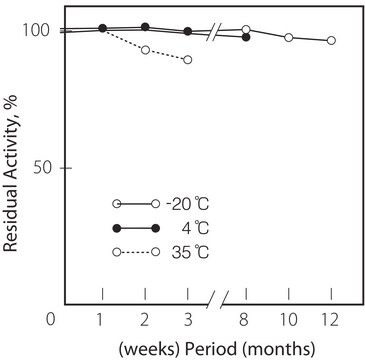
temperatura de armazenamento
−20°C
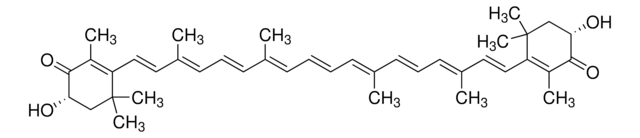
cadeia de caracteres SMILES
[H]C(=O)\C(C)=C\C=C\C(C)=C\C=C\C=C(C)\C=C\C=C(C)\C=C\C1=C(C)CCCC1(C)C
InChI
1S/C30H40O/c1-24(13-8-9-14-25(2)16-11-18-27(4)23-31)15-10-17-26(3)20-21-29-28(5)19-12-22-30(29,6)7/h8-11,13-18,20-21,23H,12,19,22H2,1-7H3/b9-8+,15-10+,16-11+,21-20+,24-13+,25-14+,26-17+,27-18+
chave InChI
DFMMVLFMMAQXHZ-DOKBYWHISA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Aplicação
- Tackling the challenge of selective analytical clean-up of complex natural extracts: the curious case of chlorophyll removal: This study outlines a methodology for the selective analytical clean-up of complex natural extracts, which can be applied to enhance the purity and stability of trans-β-Apo-8′-carotenal, particularly useful in the context of life science manufacturing and research and development. This approach is crucial for maintaining the integrity of bioactive compounds during synthesis and storage (Bijttebier et al., 2014).
Ações bioquímicas/fisiológicas
Outras notas
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 2
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica