447714
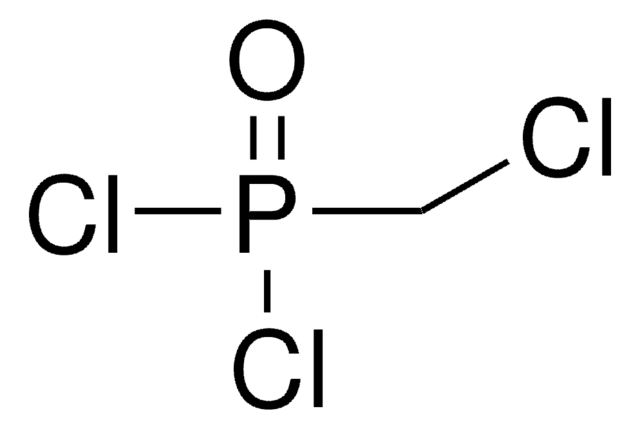
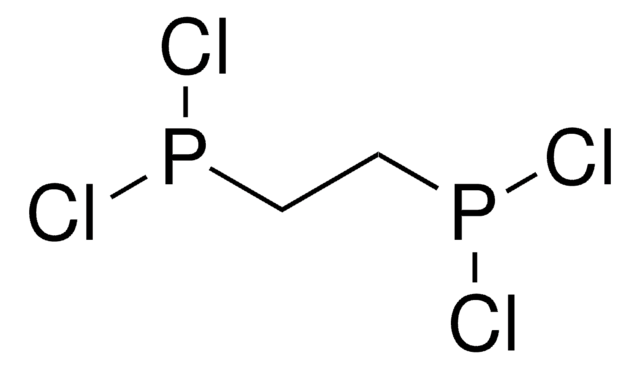
Methylenebis(phosphonic dichloride)
97%
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula linear:
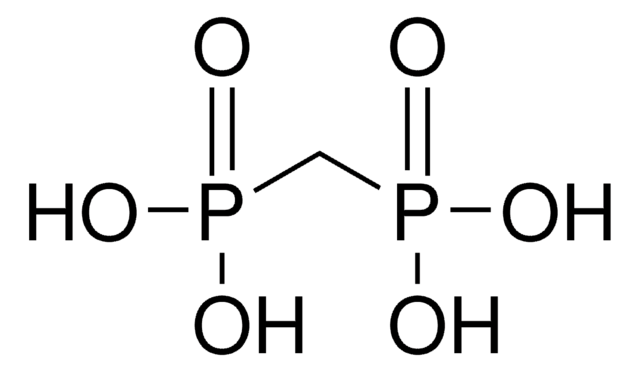
CH2[P(O)Cl2]2
Número CAS:
Peso molecular:
249.78
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Nível de qualidade
Ensaio
97%
Formulário
solid
pf
102-104 °C (lit.)
grupo funcional
phosphine oxide
cadeia de caracteres SMILES
ClP(Cl)(=O)CP(Cl)(Cl)=O
InChI
1S/CH2Cl4O2P2/c2-8(3,6)1-9(4,5)7/h1H2
chave InChI
VRXYCDTWIOCJBH-UHFFFAOYSA-N
Categorias relacionadas
Descrição geral
Methylenebis(phosphonic dichloride) is an organophosphorus compound that is commonly used in phosphonylation reactions. It is more reactive and the rate of reaction is faster compared to POCl3. This is because the phosphorus center is more electrophilic due to the lack of electron back-donation from the CH2 group.
Aplicação
Methylenebis(phosphonic dichloride) may be used for the following studies:
- Synthesis of mycophenolic methylenebis(phosphonate) derivatives.
- Phosphonylation of nucleosides.
- Preparation of P,P′-partial esters of methylenebisphosphonic acid.
- Synthesis of symmetrical di- and tetra- esters of methylenebisphosphonic acid.
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Eye Dam. 1 - Skin Corr. 1B
Perigos de suplementos
Código de classe de armazenamento
8A - Combustible corrosive hazardous materials
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Krzysztof W Pankiewicz et al.
Journal of medicinal chemistry, 45(3), 703-712 (2002-01-25)
Novel mycophenolic adenine dinucleotide (MAD) analogues have been prepared as potential inhibitors of inosine monophosphate dehydrogenase (IMPDH). MAD analogues resemble nicotinamide adenine dinucleotide binding at the cofactor binding domain of IMPDH; however, they cannot participate in hydride transfer and therefore
Facile high yielding synthesis of symmetric esters of methylenebisphosphonic acid.
Stepinski DC, et al.
Tetrahedron, 57(41), 8637-8645 (2001)
A direct method for the synthesis of nucleoside 5'-methylenebis (phosphonate) s from nucleosides.
Kalek M, et al.
Tetrahedron Letters, 46(!4), 2417-2421 (2005)
Aviran Amir et al.
The Journal of organic chemistry, 78(2), 270-277 (2012-12-05)
A new transformation of methylene-bis(phosphonic dichloride) into tetrathiobisphosphonate derivatives is reported. The reaction of methylene-bis(phosphonic dichloride) with 1,2-ethanedithiol in bromoform in the presence of AlCl(3) formed methylene-bis(1,3,2-dithiaphospholane-2-sulfide), which gave rise to O,O'-diester-methylenediphosphonotetrathioate analogues 1a-k upon reaction with phenols and alkyl
Sanjay Bhattarai et al.
Journal of medicinal chemistry, 63(6), 2941-2957 (2020-02-12)
CD73 inhibitors are promising drugs for the (immuno)therapy of cancer. Here, we present the synthesis, structure-activity relationships, and cocrystal structures of novel derivatives of the competitive CD73 inhibitor α,β-methylene-ADP (AOPCP) substituted in the 2-position. Small polar or lipophilic residues increased
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica