438537
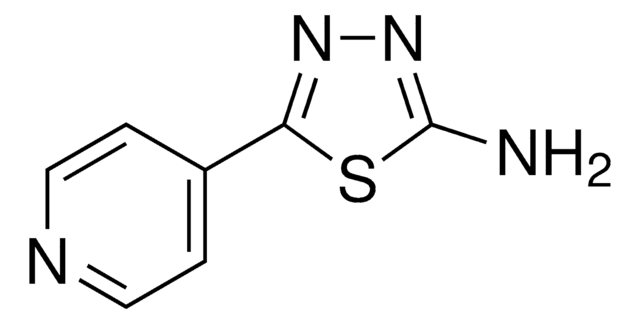
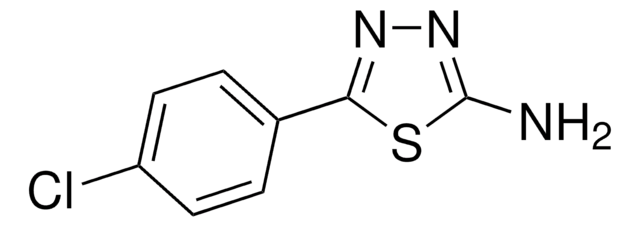
2-Amino-5-phenyl-1,3,4-thiadiazole
96%
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula empírica (Notação de Hill):
C8H7N3S
Número CAS:
Peso molecular:
177.23
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Nível de qualidade
Ensaio
96%
Formulário
solid
pf
223-227 °C (lit.)
grupo funcional
phenyl
cadeia de caracteres SMILES
Nc1nnc(s1)-c2ccccc2
InChI
1S/C8H7N3S/c9-8-11-10-7(12-8)6-4-2-1-3-5-6/h1-5H,(H2,9,11)
chave InChI
UHZHEOAEJRHUBW-UHFFFAOYSA-N
Categorias relacionadas
Descrição geral
2-Amino-5-phenyl-1,3,4-thiadiazole on condensation with benzaldehyde (SPT), 4-nitrobenzaldehyde (SNT), 4-methoxybenzaldehyde (SMT), 2-hydroxybenzaldehyde (SSTH) or 2-hydroxyacetophenone affords Schiff′s bases. Its molecular geometry and vibrational frequencies have been evaluated using the Hartree-Fock and density functional method (B3LYP). 2-Amino-5-phenyl-1,3,4-thiadiazole has been reported to inhibit the corrosion of mild steel in 0.5M H2SO4 and 1.0M HCl. Corrosion inhibition has been examined using potentiodynamic polarization and electrochemical impedance spectroscopy (EIS).
Aplicação
2-Amino-5-phenyl-1,3,4-thiadiazole may be used for the synthesis of 1,3,4-thiadiazole derivatives.
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órgãos-alvo
Respiratory system
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Equipamento de proteção individual
dust mask type N95 (US), Eyeshields, Gloves
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Synthesis, characterization and antimicrobial activity of some carbamothioyl-1, 3, 4-thiadiazole derivatives.
Salih N, et al.
Synthesis, 4(2), 655-660 (2012)
A K Srivastava et al.
Bioinorganic chemistry and applications, 3(3-4), 289-297 (2008-03-28)
The reactions of bis(cyclopentadienyl)titanium(IV) dichloride with Schiff bases derived by condensing 2- amino-5-phenyl-1,3,4-thiadiazole with benzaldehyde (SPT), 4-nitrobenzaldehyde (SNT), 4-methoxybenzaldehyde (SMT), 2-hydroxybenzaldehyde (SSTH) or 2-hydroxyacetophenone (SATH) have been studied in refluxing tetrahydrofuran and complexes of types [Cp(2)TiCl(SB)]Cl (SB= SPT, SNT or
Experimental and molecular dynamics studies on corrosion inhibition of mild steel by 2-amino-5-phenyl-1, 3, 4-thiadiazole.
Tang Y, et al.
Corrosion Science, 52(1), 242-249 (2010)
Y Atalay et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 64(1), 68-72 (2005-08-13)
The molecular geometry and vibrational frequencies of 2-amino-5-phenyl-1,3,4-thiadiazole (C8H7N3S) in the ground state has been calculated using the Hartree-Fock and density functional method (B3LYP) with 6-31G(d) basis set. The optimized geometric bond lengths and bond angles obtained by using HF
Jingjing Zhang et al.
Carbohydrate polymers, 215, 108-118 (2019-04-15)
In the current study, five novel urea-functionalized chitosan derivatives were synthesized via condensation reactions of chloroacetyl chitosan (CTCS) with urea groups bearing nitrogen-containing heterocycles. In order to identify the structure characteristics of chitosan derivatives, FT-IR, 1H NMR spectroscopy, and elemental
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica