About This Item
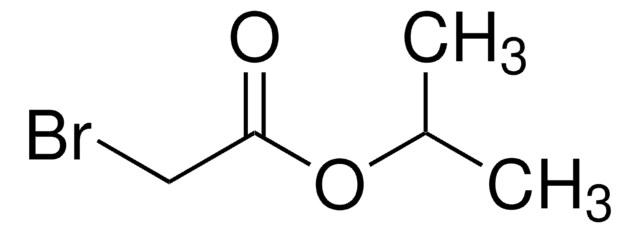
Fórmula linear:
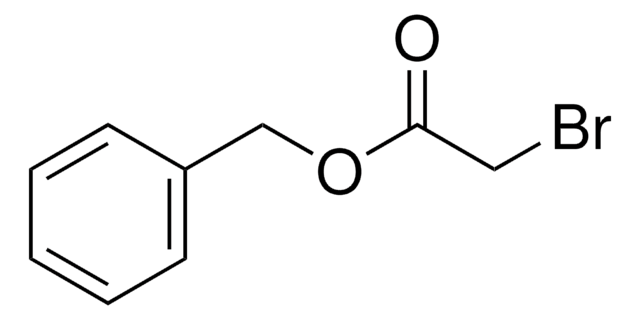
BrCH2CO2C6H5
Número CAS:
Peso molecular:
215.04
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Ensaio
98%
Formulário
solid
p.e.
134 °C/15 mmHg (lit.)
pf
31-33 °C (lit.)
densidade
1.508 g/mL at 25 °C (lit.)
grupo funcional
bromo
ester
phenoxy
cadeia de caracteres SMILES
BrCC(=O)Oc1ccccc1
InChI
1S/C8H7BrO2/c9-6-8(10)11-7-4-2-1-3-5-7/h1-5H,6H2
chave InChI
UEWYUCGVQMZMGY-UHFFFAOYSA-N
Descrição geral
Phenyl bromoacetate is an aromatic ester.
Aplicação
Phenyl bromoacetate may be employed as alkylation reagent in the preparation of 2-(phenoxycarbonyl)methyl triazoles. It may be used in the synthesis of the A-ring of cylindrospermopsin. It may be used in the synthesis of the following 4-thiazolidinones:
- 2-{2-[1-(4-hydroxy-6-methyl-2-oxo-2H-pyran-3-yl)ethylidene]hydrazinyl}-1,3-thiazolidin-4-one
- 2-{2-[1-(4-hydroxy-6-methyl-2-oxo-2H-pyran-3-yl)ethylidene]hydrazinyl}-3-methyl-1,3-thiazolidin-4-one
- 3-ethyl-2-{2-[1-(4-hydroxy-6-methyl-2-oxo-2H-pyran-3-yl)ethylidene]hydrazinyl}-1,3-thiazolidin-4-one
- 2-{2-[1-(4-hydroxy-6-methyl-2-oxo-2H-pyran-3-yl)ethylidene] hydrazinyl}-3-phenyl-1,3-thiazolidin-4-one
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Synthesis and Antimicrobial Activity Evaluation of Novel 4-Thiazolidinones Containing a Pyrone Moiety.
Nechak R, et al.
Synthetic Communications, 1-11 (2014)
Robert M Williams et al.
ACS symposium series. American Chemical Society, 1009, 420-442 (2010-06-22)
We report the application of diphenyloxazinone glycinate chiral templates to asymmetric syntheses of cylindrospermospin, 7-epi-cylindrospermopsin, 7-deoxycylindrospermopsin, and spirotryprostatins A and B. Synthetic studies toward quinine, nakadomarin A, and palau'amine using these templates are also described.
Duen-Ren Hou et al.
Bioorganic & medicinal chemistry letters, 19(3), 1022-1025 (2008-12-20)
This letter reports the new entry of novel 1,2,3-triazole derivatives as CB1 receptor antagonists. The design, synthesis and biological evaluation of N1 and N2 substituted 1,2,3-trizoles are described. The N2 substituted, symmetrical 1,2,3-triazoles are more potent ligands than the unsymmetrical
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica