156671
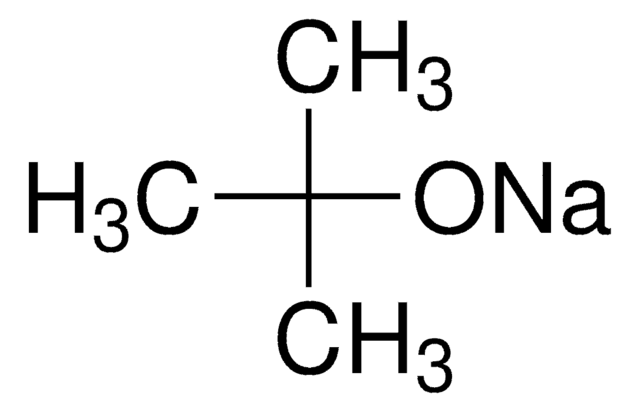
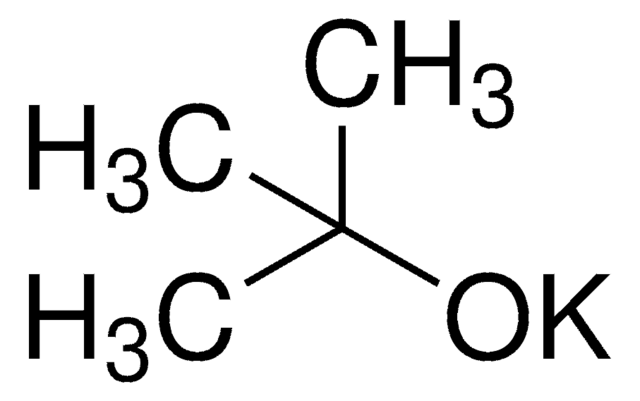
Potassium tert-butoxide
reagent grade, ≥98%
Sinônimo(s):
Potassium tert-butylate, Potassium t-butoxide
About This Item
Produtos recomendados
grau
reagent grade
Nível de qualidade
pressão de vapor
1 mmHg ( 220 °C)
Ensaio
≥98%
Formulário
solid
características do produto alternativo mais ecológico
Catalysis
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
pf
256-258 °C (dec.) (lit.)
categoria alternativa mais ecológica
cadeia de caracteres SMILES
[K+].CC(C)(C)[O-]
InChI
1S/C4H9O.K/c1-4(2,3)5;/h1-3H3;/q-1;+1
chave InChI
LPNYRYFBWFDTMA-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Descrição geral
Aplicação
It can also be used:
- To synthesize aliphatic and aromatic amides from corresponding esters and amines.
- As a base in the intramolecular cyclization of aryl ethers, amines, and amides.
- As a catalyst to prepare styrene derivatives from aryl halides and alkenes by Mizoroki-Heck reaction.
tert-Butoxide-Assisted Amidation of Esters under Green Conditions
Potassium tert-butoxide may be used as a base in the intramolecular cyclization of iodo arene to afford benzopyran via microwave method of synthesis.
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Eye Dam. 1 - Flam. Sol. 1 - Self-heat. 2 - Skin Corr. 1A
Perigos de suplementos
Código de classe de armazenamento
4.2 - Pyrophoric and self-heating hazardous materials
Classe de risco de água (WGK)
WGK 1
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Protocolos
Information on the Amide bond and the Catalytic Amide Bond Formation Protocol. Amidation of amines and alcohols. The amide bond, an important linkage in organic chemistry, is a key functional group in peptides, polymers, and many natural products and pharmaceuticals.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica