137561
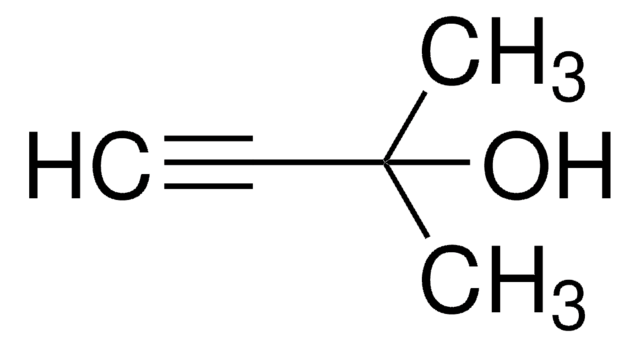
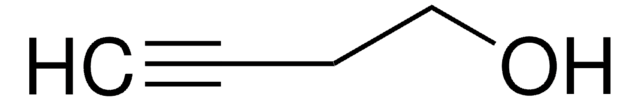
3-Methyl-1-pentyn-3-ol
98%
Sinônimo(s):
Ethyl ethynyl methyl carbinol, Meparfynol
About This Item
Produtos recomendados
densidade de vapor
3 (vs air)
pressão de vapor
6.5 mmHg ( 20 °C)
Ensaio
98%
Formulário
liquid
índice de refração
n20/D 1.431 (lit.)
p.e.
121-122 °C (lit.)
solubilidade
Cellosolve: miscible
Stoddard solvent: miscible
acetone: miscible
benzene: miscible
carbon tetrachloride: miscible
cyclohexanone: miscible
diethyl ether: soluble
diethylene glycol: miscible
ethanolamine: miscible
ethyl acetate: miscible
kerosene: miscible
mineral spirits: miscible
neatsfoot oil: miscible
petroleum ether: miscible
soybean oil: miscible
densidade
0.866 g/mL at 25 °C (lit.)
grupo funcional
hydroxyl
cadeia de caracteres SMILES
CCC(C)(O)C#C
InChI
1S/C6H10O/c1-4-6(3,7)5-2/h1,7H,5H2,2-3H3
chave InChI
QXLPXWSKPNOQLE-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
Aplicação
- A reactant to synthesize α-methylene cyclic carbonates by reacting with carbon dioxide.
- A reactant in the synthesis of 2,6,9-trisubstituted purine based CDK inhibitors.
- An initiator in the synthesis of polylactide bearing terminal propargyl group via ring-opening polymerization of L-lactide.
Ações bioquímicas/fisiológicas
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Acute Tox. 4 Oral - Aquatic Chronic 3 - Eye Dam. 1 - Flam. Liq. 3
Código de classe de armazenamento
3 - Flammable liquids
Classe de risco de água (WGK)
WGK 2
Ponto de fulgor (°F)
82.4 °F - closed cup
Ponto de fulgor (°C)
28 °C - closed cup
Equipamento de proteção individual
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica