Key Documents
SML2368
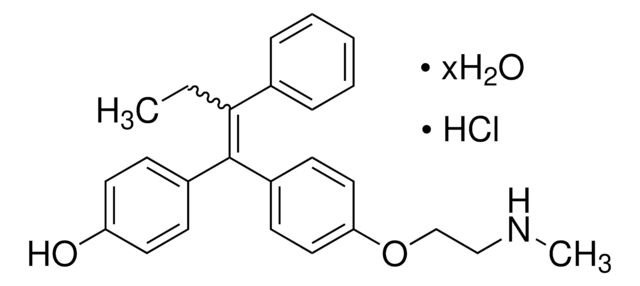
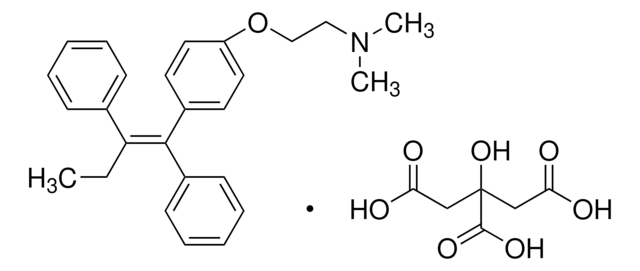
(Z)-Endoxifen
≥98% (HPLC)
Synonim(y):
4-Hydroxy-N-desmethyl-tamoxifen, Endoxifen, Metabolite BX, N-Desmethyl-4-hydroxytamoxifen, Z-Endoxifen
About This Item
Polecane produkty
Próba
≥98% (HPLC)
Postać
powder
kolor
white to beige
rozpuszczalność
DMSO: 2 mg/mL, clear
temp. przechowywania
2-8°C
Zastosowanie
Działania biochem./fizjol.
Hasło ostrzegawcze
Danger
Zwroty wskazujące rodzaj zagrożenia
Zwroty wskazujące środki ostrożności
Klasyfikacja zagrożeń
Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 1A - Repr. 1B
Kod klasy składowania
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej