Key Documents
SML1929
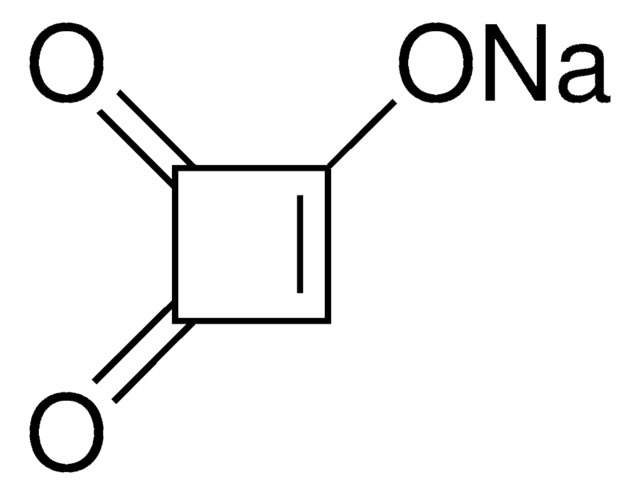
Equisetin
(Fusarium equiseti), ≥97% (HPLC)
Synonim(y):
(3E,5S)-3-[[(1S,2R,4aS,6R,8aR)-1,6-dimethyl-2-[(E)-prop-1-enyl]-4a,5,6,7,8,8a-hexahydro-2H-naphthalen-1-yl]-hydroxymethylidene]-5-(hydroxymethyl)-1-methylpyrrolidine-2,4-dione
About This Item
Polecane produkty
Poziom jakości
pochodzenie biologiczne
(Fusarium equiseti)
Próba
≥97% (HPLC)
Postać
powder
rozpuszczalność
DMSO: soluble 1 mg/mL
chloroform: soluble 5 mg/mL
Warunki transportu
ambient
temp. przechowywania
−20°C
ciąg SMILES
C[C@H]1C[C@]2([H])[C@@]([C@](C)(/C(O)=C3C([C@H](CO)N(C)C/3=O)=O)[C@H](/C=C/C)C=C2)([H])CC1
InChI
1S/C22H31NO4/c1-5-6-15-9-8-14-11-13(2)7-10-16(14)22(15,3)20(26)18-19(25)17(12-24)23(4)21(18)27/h5-6,8-9,13-17,24,27H,7,10-12H2,1-4H3/b6-5+/t13-,14-,15-,16-,17+,22-/m1/s1
Klucz InChI
SLOQQJFOWFUUMP-KNQICWOTSA-N
Działania biochem./fizjol.
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej

![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)