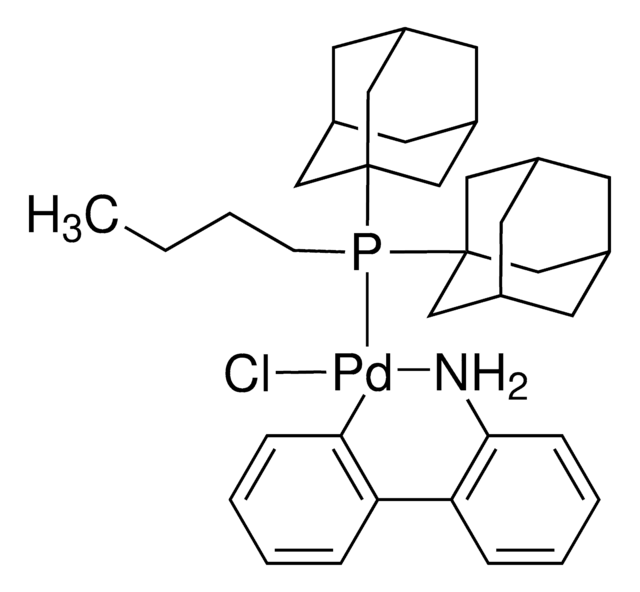
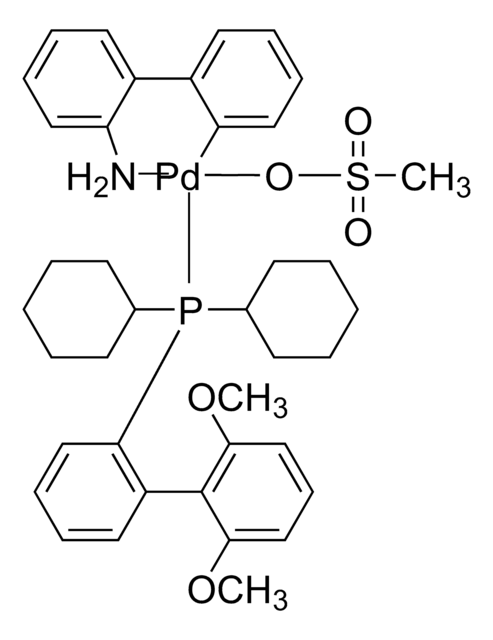
Kluczowe dokumenty
732117
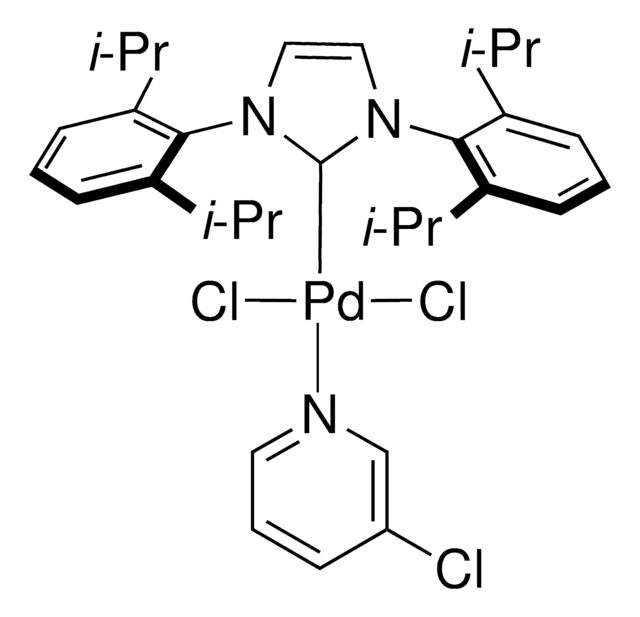
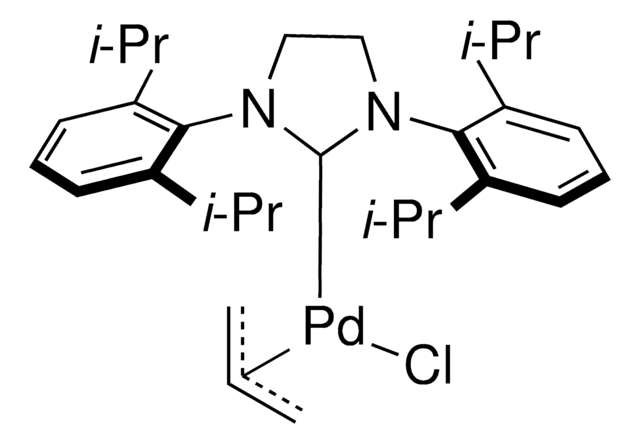
Pd-PEPPSI™-IPent catalyst
≥95%
Synonim(y):
Dichloro[1,3-bis(2,6-Di-3-pentylphenyl)imidazol-2-ylidene](3-chloropyridyl)palladium(II), [1,3-Bis(2,6-Di-3-pentylphenyl)imidazol-2-ylidene](3-chloropyridyl)dichloropalladium(II), [1,3-Bis(2,6-Di-3-pentylphenyl)imidazol-2-ylidene](3-chloropyridyl)palladium(II) dichloride
About This Item
Polecane produkty
Poziom jakości
Próba
≥95%
Formularz
solid
przydatność reakcji
core: palladium
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Cross Couplings
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: catalyst
mp
195-201 °C
temp. przechowywania
−20°C
ciąg SMILES
Clc1cccnc1.CCC(CC)c2cccc(C(CC)CC)c2N3C=CN(c4c(cccc4C(CC)CC)C(CC)CC)\C3=[Pd](/Cl)Cl
InChI
1S/C35H52N2.C5H4ClN.2ClH.Pd/c1-9-26(10-2)30-19-17-20-31(27(11-3)12-4)34(30)36-23-24-37(25-36)35-32(28(13-5)14-6)21-18-22-33(35)29(15-7)16-8;6-5-2-1-3-7-4-5;;;/h17-24,26-29H,9-16H2,1-8H3;1-4H;2*1H;/q;;;;+2/p-2
Klucz InChI
BCXSKTXOKALLAZ-UHFFFAOYSA-L
Opis ogólny
Zastosowanie
Cross-Coupling, Amination and Heck Transformation using PEPPSI Catalysts
- Catalyst for Stille coupling reaction (eq. 1)
- Catalyst for Negishi coupling reaction (eq. 2)
- Catalyst for Suzuki coupling reaction (eq. 3)
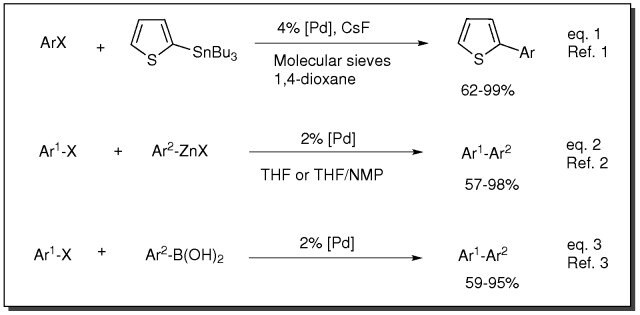
For small scale and high throughput uses, product is also available as ChemBeads (928399)
Informacje prawne
produkt powiązany
Hasło ostrzegawcze
Warning
Zwroty wskazujące rodzaj zagrożenia
Zwroty wskazujące środki ostrożności
Klasyfikacja zagrożeń
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Produkty
Professor Mike Organ and co-workers have developed the PEPPSI™ (Pyridine-Enhanced Precatalyst Preparation Stabilization and Initiation) precatalysts for palladium-catalyzed cross-coupling reactions.
Professor Mike Organ at York University, along with co-workers Dr. Chris O’Brien and Dr. Eric Kantchev, have developed an palladium N-heterocyclic-carbene (NHC) catalyst system. They reacted PdCl2with a bulky NHC ligand, 2,6-diisopropylphenyllimidazolium chloride (IPr), and an α-donating 3-chloropyridine ligand for stability. The title complex, PEPPSI™, stands for Pyridine-Enhanced Precatalyst Preparation Stabilization and Initiation. Sigma-Aldrich offesr gram-scale quantities of the PEPPSI™ catalyst in collaboration with the Organ research group.
All of the preformed catalysts used in the kit are air and moisture stable complexes in their commercially available form.
Multiple tools have been created to ensure your success with kit set up. Start with the more detailed guide to ensure you are comfortable with all of the steps before using the quick guides on the excel worksheet. Remember that while the technique is new, it is still organic chemistry and so the steps will seem easy once you try just one kit. It is just a new way of approaching something you are already very good at.
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej