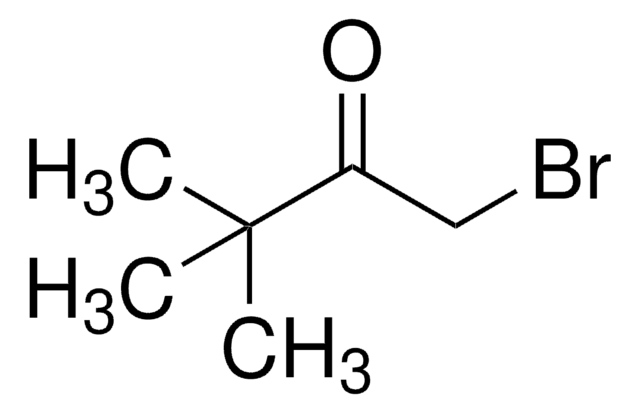
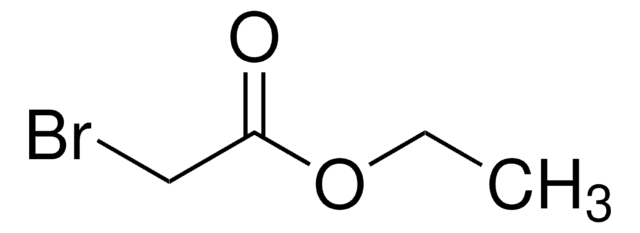
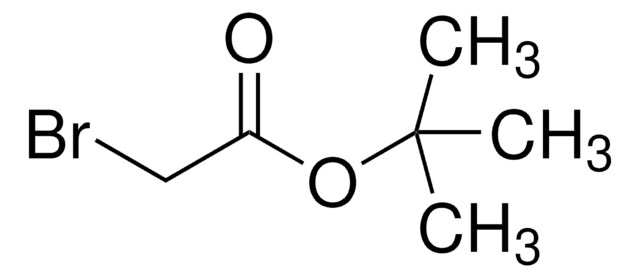
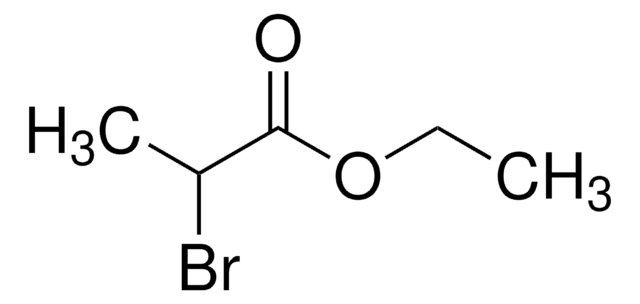
おすすめの製品
詳細
1-Bromopinacolone is an α-bromoketone-aldehyde. Coupling of 1-bromopinacolone with various aldehyde electrophiles catalyzed by SmI2 has been reported. It acts as reversible competitive inhibitor for acetylcholinesterase, during hydrolysis of acetylcholine.
アプリケーション
1-Bromopinacolone is the suitable reagent for the synthesis of a photolabile azido derivative of the kaurene oxidase inhibitor 1-(4- chlorophenyl)4,4-dimethyl-2-(1,2,4-triazol-1-yl) pentan-3-ol (paclobutrazol). It may be used as reagent in the synthesis of 2-t-butyl-6-benzoylimidazo[1,2-b]pyridazine.
シグナルワード
Warning
危険有害性情報
危険有害性の分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
ターゲットの組織
Respiratory system
保管分類コード
10 - Combustible liquids
WGK
WGK 3
引火点(°F)
167.0 °F - closed cup
引火点(℃)
75 °C - closed cup
個人用保護具 (PPE)
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
消防法
第4類:引火性液体
第三石油類
危険等級III
非水溶性液体
Jan Code
414131-25ML:
414131-BULK:
414131-VAR:
414131-5ML:
この製品を見ている人はこちらもチェック
Brian A Sparling et al.
Organic letters, 10(6), 1291-1294 (2008-02-28)
Highly substituted, very hindered enones were synthesized using a two-step procedure that utilizes a diiodosamarium-promoted Reformatsky-type coupling and dehydration using Martin sulfurane. Both alpha-chloro- and alpha-bromoketones were coupled with a variety of carbonyl nucleophiles to form the intermediate beta-hydroxyketones, occurring
Methyl imidazo [1,2-b] pyridazine-2-carbamates and related compounds as potential antifilarial agents
Mourad AE, et al.
Journal of Heterocyclic Chemistry, 29, 1583-1583 (1992)
D L Hallahan et al.
Plant physiology, 88(4), 1425-1429 (1988-12-01)
A photolabile azido derivative of the kaurene oxidase inhibitor 1-(4-chlorophenyl)-4,4-dimethyl-2-(1,2,4-triazol-l-yl) pentan-3-ol (paclobutrazol) has been synthesized for use as a photoaffinity labeling agent. The compound was tested as an inhibitor of the oxidation of ent-kaurene catalyzed by cell-free preparations from endosperm
S G Cohen et al.
The Journal of biological chemistry, 257(23), 14087-14092 (1982-12-10)
1-Bromopinacolone, BrPin, acts initially as a reversible competitive inhibitor for acetylcholinesterase, KI = 0.18 mM in hydrolysis of acetylcholine. Unlike bromoacetone, with time it acts as an irreversible covalent inhibitor. BrPin has a hydrolytic half-life of 30 h at the
S G Cohen et al.
Biochimica et biophysica acta, 997(3), 167-175 (1989-08-31)
1-Bromo-2-[14C]pinacolone, (CH3)3C14COCH2Br [( 14C]BrPin), was prepared from [1-14C]acetyl chloride and tert-butylmagnesium chloride with cuprous chloride catalyst, followed by bromination. It was examined as an active-site directed label for acetylcholinesterase (acetylcholine acetylhydrolase, EC 3.1.1.7) (AcChE). AcChE, isolated from Torpedo nobiliana, has
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)