903000
[4,4′-Bis(1,1-dimethylethyl)-2,2′-bipyridine] nickel (II) dichloride
Sinonimo/i:
(4,4′-dtbbpy)NiCl2
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C18H24Cl2N2Ni
Numero CAS:
Peso molecolare:
398.00
Numero MDL:
Codice UNSPSC:
12352101
NACRES:
NA.22
Prodotti consigliati
Stato
powder or crystals
Impiego in reazioni chimiche
core: nickel
reaction type: Cross Couplings
reagent type: catalyst
Punto di fusione
>300 °C
Stringa SMILE
CC(C1=CC(C2=CC(C(C)(C)C)=CC=N2)=NC=C1)(C)C.Cl[Ni]Cl
InChI
PCWIKFRTCXESOT-UHFFFAOYSA-L
Applicazioni
[4,4′-Bis(1,1-dimethylethyl)-2,2′-bipyridine] nickel (II) dichloride can be used as a catalyst in:
- Decarboxylative arylation of oxo acids.
- Acylation of ethers.
- Cross-coupling of aryl bromides with alcohols.
Prodotti correlati
N° Catalogo
Descrizione
Determinazione del prezzo
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
Lot/Batch Number
Non trovi la versione di tuo interesse?
Se hai bisogno di una versione specifica, puoi cercare il certificato tramite il numero di lotto.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Wacharee Harnying et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 17(17), 4765-4773 (2011-03-23)
The roles of nickel and chromium catalysts in the coupling reaction of vinyl halides and aldehydes, the so-called Nozaki-Hiyama-Kishi (NHK) reaction, have been studied by UV/Vis spectroscopy, electrochemical, and spectroelectrochemical methods. Electrochemical studies revealed that nickel plays the central role
Lingling Chu et al.
Angewandte Chemie (International ed. in English), 54(27), 7929-7933 (2015-05-28)
The direct decarboxylative arylation of α-oxo acids has been achieved by synergistic visible-light-mediated photoredox and nickel catalysis. This method offers rapid entry to aryl and alkyl ketone architectures from simple α-oxo acid precursors via an acyl radical intermediate. Significant substrate
Photocatalytic α-Acylation of Ethers
Sun Z, et al.
Organic Letters, 19, 3727-3730 (2017)
Martins S Oderinde et al.
The Journal of organic chemistry, 80(15), 7642-7651 (2015-07-04)
In order to achieve reproducibility during iridium-photoredox and nickel dual-catalyzed sp(3)-sp(2) carbon-carbon bond-forming reactions, we investigated the role that molecular oxygen (O2), solvent and light-source (CF lamp or blue LED) play in a variety of Ir-photoredox mediated transformations. The presence
Aryl Ketones as Single-Electron-Transfer Photoredox Catalysts in Nickel-Catalyzed the Homocoupling of Aryl Halides
Masuda Y, et al.
European Journal of Organic Chemistry, 5822-5825 (2016)
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.![(Ir[dF(CF3)ppy]2(dtbpy))PF6](/deepweb/assets/sigmaaldrich/product/structures/982/913/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09/640/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09.png)
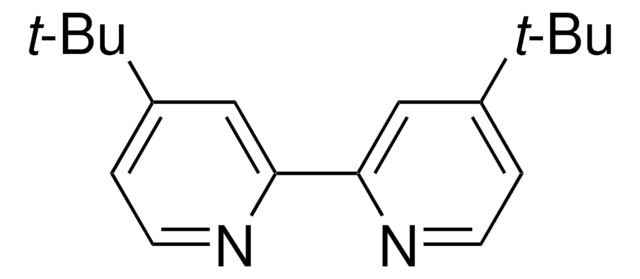

![[Ni(dtbbpy)(H2O)4]Cl2](/deepweb/assets/sigmaaldrich/product/structures/777/629/15c13300-e874-4abd-8bd4-8b2bb4864570/640/15c13300-e874-4abd-8bd4-8b2bb4864570.png)
![[Ir(dtbbpy)(ppy)2]PF6](/deepweb/assets/sigmaaldrich/product/structures/158/329/2544d673-d267-4aa1-8f46-2652aad4bfa0/640/2544d673-d267-4aa1-8f46-2652aad4bfa0.png)
![[Ir(dF(Me)ppy)2(dtbbpy)]PF6](/deepweb/assets/sigmaaldrich/product/structures/150/099/7c2dfa31-39f4-4cca-aee5-86d4a89fea78/640/7c2dfa31-39f4-4cca-aee5-86d4a89fea78.png)
![Bis[(2-dimethylamino)phenyl]amine nickel(II) chloride ≥97% (AT)](/deepweb/assets/sigmaaldrich/product/structures/143/670/3d0cc911-c810-4324-914e-85c5c11b7dac/640/3d0cc911-c810-4324-914e-85c5c11b7dac.png)
![[(TMEDA)Ni(o-tolyl)Cl] 95%](/deepweb/assets/sigmaaldrich/product/structures/236/439/768c916e-994f-47e3-a980-3ca0471317d7/640/768c916e-994f-47e3-a980-3ca0471317d7.png)
![[Ir{dFCF3ppy}2(bpy)]PF6](/deepweb/assets/sigmaaldrich/product/structures/180/924/79119ac4-7d62-429d-b23d-a14c012c6050/640/79119ac4-7d62-429d-b23d-a14c012c6050.png)
![[1,3-Bis(diphenylphosphino)propane]dichloronickel(II)](/deepweb/assets/sigmaaldrich/product/structures/844/065/af07f787-c6a3-4a6e-a22b-47a933c73978/640/af07f787-c6a3-4a6e-a22b-47a933c73978.png)