K4769
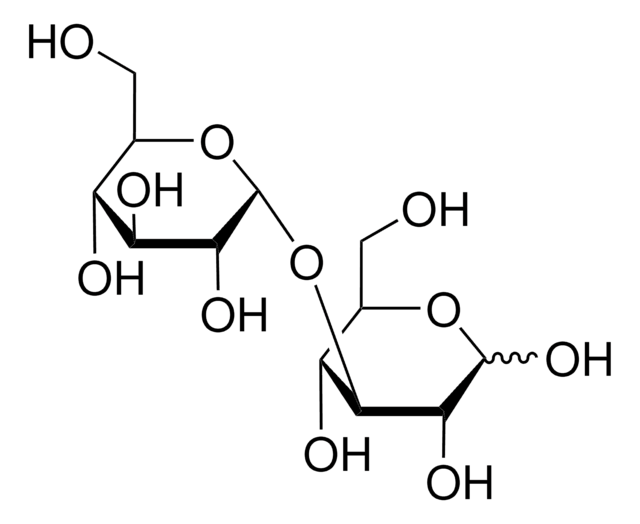
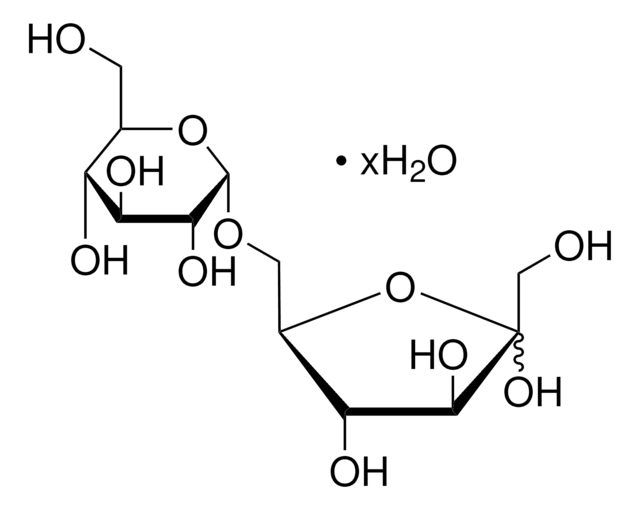
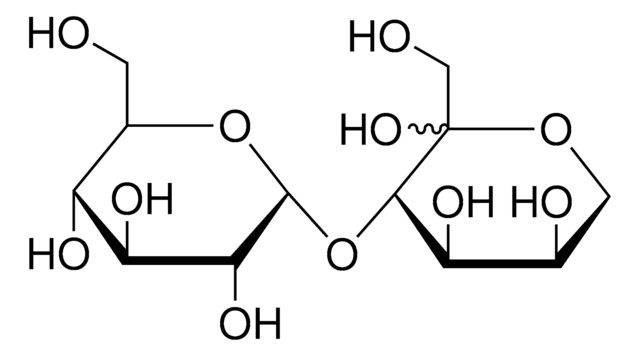
Kojibiose
≥98% (HPLC)
Sinônimo(s):
α-D-Glc-(1→2)-D-Glc, 2-O-α-D-Glucopyranosyl-D-glucose
About This Item
Produtos recomendados
Nível de qualidade
Ensaio
≥98% (HPLC)
Formulário
powder
técnica(s)
HPLC: suitable
cor
white to off-white
solubilidade
water: 5 mg/mL, clear, colorless
temperatura de armazenamento
−20°C
cadeia de caracteres SMILES
OCC(O)C(O)C(O)C(OC1OC(CO)C(O)C(O)C1O)C=O
InChI
1S/C12H22O11/c13-1-4(16)7(17)8(18)5(2-14)22-12-11(21)10(20)9(19)6(3-15)23-12/h2,4-13,15-21H,1,3H2
chave InChI
PZDOWFGHCNHPQD-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Aplicação
Ações bioquímicas/fisiológicas
Outras notas
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica