D9150
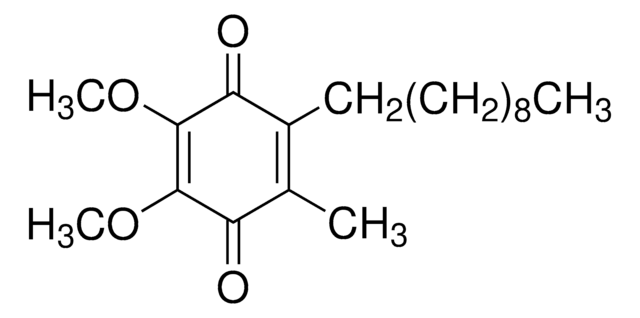
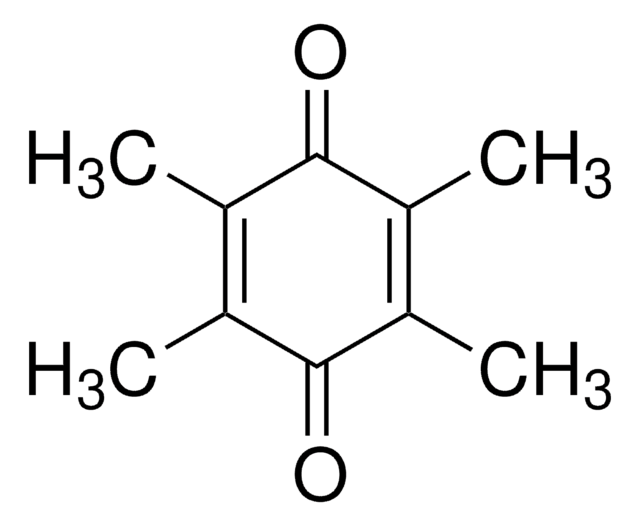
2,3-Dimethoxy-5-methyl-p-benzoquinone
apoptosis inducer
Sinônimo(s):
Coenzyme Q0
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula empírica (Notação de Hill):
C9H10O4
Número CAS:
Peso molecular:
182.17
Beilstein:
1640422
Número CE:
Número MDL:
Código UNSPSC:
12352204
ID de substância PubChem:
NACRES:
NA.51
Produtos recomendados
Formulário
powder
pf
58-60 °C (lit.)
temperatura de armazenamento
2-8°C
cadeia de caracteres SMILES
COC1=C(OC)C(=O)C(C)=CC1=O
InChI
1S/C9H10O4/c1-5-4-6(10)8(12-2)9(13-3)7(5)11/h4H,1-3H3
chave InChI
UIXPTCZPFCVOQF-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Descrição geral
2,3-Dimethoxy-5-methyl-p-benzoquinone (Coenzyme Q0 or DMM) is present in all the cells including neural cells.
Aplicação
2,3-Dimethoxy-5-methyl-p-benzoquinone has been used:
- as a tau protein fibrillization inducer to determine the regions of tau involved in the formation of paired helical filaments (PHFs)
- as a component in buffer B for cytochrome oxidation assay with subsaturating light
- in the RPMI-1640 medium for 2,3-bis-(2-methoxy-4-nitro-5-sulphenyl)-(2H)-tetrazolium-5-carboxanilide (XTT) assay to quantify antifungal activity
Coenzyme Q0 inhibits (via radical quenching) reactions of gamma-irradiation induced homolytic cleavage of O-glycoside bonds in polysaccharides. Coenzyme Q0 induces apoptosis and modulates the cell cycle in estrogen receptor negative breast cancer cells. It is toxic to other cells such as insulin producing cells.
Ações bioquímicas/fisiológicas
2,3-Dimethoxy-5-methyl-p-benzoquinone (Coenzyme Q0) interacts with tau protein and aids in the formation of filamentous structure.
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órgãos-alvo
Respiratory system
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 2
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
dust mask type N95 (US), Eyeshields, Gloves
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
XTT assay of antifungal activity
Loures FV and Levitz SM
PLoS Pathogens, 5(15), e1543-e1543 (2015)
The Photosynthetic Bacterial Reaction Center: Structure and Dynamics, 114-114 (2013)
Augustin C Mot et al.
PloS one, 15(1), e0225530-e0225530 (2020-01-22)
Yellow laccases lack the typical blue type 1 Cu absorption band around 600 nm; however, multi-copper oxidases with laccase properties have been reported. We provide the first evidence that the yellow laccase isolated from Sclerotinia sclerotiorum is obtained from a
In vitro tau fibrillization: mapping protein regions
Santa-Maria I, et al.
Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease, 1762(7), 683-692 (2006)
Yung-Fu Wang et al.
Bioelectrochemistry (Amsterdam, Netherlands), 69(1), 74-81 (2006-01-25)
Bioelectrocatalytic oxidation of acetate was investigated under anaerobic conditions by using Escherichia coli K-12 (IFO 3301) cells cultured on aerobic media containing poly-peptone, glucose or acetate as the sole carbon source. It was found that all E. coli cells cultured
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica