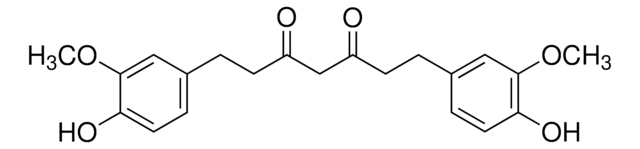
B6938
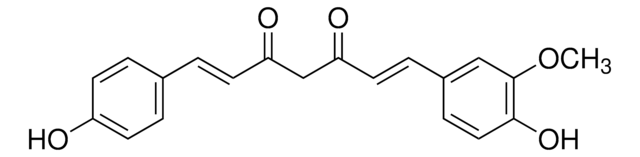
Bisdemethoxycurcumin
≥98% (HPLC), solid
Sinônimo(s):
(1E,6E)-1,7-bis(4-hydroxyphenyl)hepta-1,6-diene-3,5-dione
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula empírica (Notação de Hill):
C19H16O4
Número CAS:
Peso molecular:
308.33
Número MDL:
Código UNSPSC:
12352205
ID de substância PubChem:
NACRES:
NA.77
Produtos recomendados
Ensaio
≥98% (HPLC)
Formulário
solid
solubilidade
DMSO: ≥20 mg/mL
temperatura de armazenamento
2-8°C
cadeia de caracteres SMILES
Oc1ccc(cc1)\C=C\C(=O)CC(=O)\C=C\c2ccc(O)cc2
InChI
1S/C19H16O4/c20-16-7-1-14(2-8-16)5-11-18(22)13-19(23)12-6-15-3-9-17(21)10-4-15/h1-12,20-21H,13H2/b11-5+,12-6+
chave InChI
PREBVFJICNPEKM-YDWXAUTNSA-N
Categorias relacionadas
Aplicação
Bisdemethoxycurcumin has been used:
- to test its inhibitory effect on cell cycle and mitochondrial function in gastric adenocarcinoma cells
- to test it neuroprotective role against lead (Pb) induced toxicity in dopaminergic and noradrenergic systems of Meriones shawi
- as a standard for calibration curve generation to quantify plasma curcuminoids using high-performance liquid chromatography-diode array detection (HPLC-DAD) and ultraviolet (UV)
Ações bioquímicas/fisiológicas
Bisdemethoxycurcumin (BDMC) is a derivative or curcumin, and represents one of the major active components of curcumin products isolated from Curcumae sp. BDMC shares similar anti-inflammatory properties with demethoxycurcumin. It inhibits LPS-induced nitric oxide (NO) production and expression of iNOS and COX2 in RAW264.7 cells by blocking NF-kB activation. BDMC also displays unique properties in that it enhances Abeta clearance by upregulating expression MGAT3 and TLR genes. BDMC potently inhibits AKR1B10.
Bisdemethoxycurcumin (BMDC) is a stable dimethoxy derivative of curcumin and is useful as a supplement in cell culture medium. It also possesses antimicrobial, antioxidative and neuroprotective functionality.
Inhibitor of NF-kB transcriptional activity
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Lahcen Tamegart et al.
Comptes rendus biologies, 342(5-6), 192-198 (2019-09-03)
Exposure to lead is a threat factor for neurodegenerative disorders progress as it could trigger dopaminergic deficiency. We aimed herein to assess the effect of acute lead exposure (25mg/kg B.W i.p.) during three continuous days on the dopaminergic and noradrenergic
Wojciech Ostrowski
European journal of mass spectrometry (Chichester, England), 21(1), 45-50 (2015-04-24)
Curcumin complexes with iron ions were investigated by electrospray ionization mass spectrometry. It was shown that in methanol solutions of curcumin and iron(III) ions, complexes are formed with a stoichiometry of 1 : 1, 2 : or 3 : 1.
Hannelore Rücker et al.
Organic & biomolecular chemistry, 13(10), 3040-3047 (2015-01-27)
Inflammatory signaling pathways orchestrate the cellular response to infection and injury. These pathways are known to be modulated by compounds that alkylate cysteinyl thiols. One class of phytochemicals with strong thiol alkylating activity is the chalcones. In this study we
Meyliana Wulandari et al.
Analytical and bioanalytical chemistry, 407(3), 803-812 (2014-07-25)
This paper describes the synthesis of novel molecularly imprinted polymers (MIPs), prepared by a noncovalent imprinting approach, for cleanup and preconcentration of curcumin (CUR) and bisdemethoxycurcumin (BDMC) from medicinal herbal extracts and further analysis by high-performance liquid chromatography with fluorescence
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica