S4921
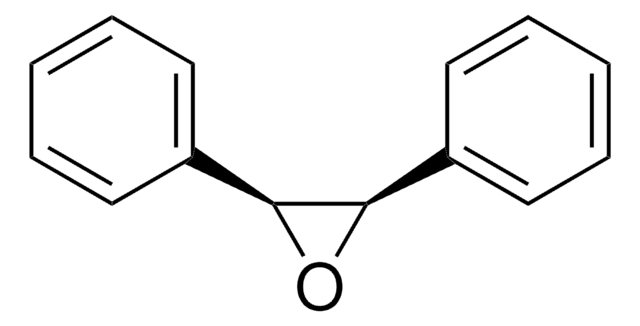
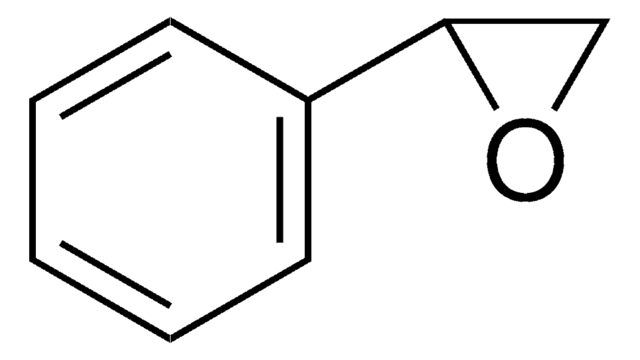
trans-Stilbene oxide
98%
Sinônimo(s):
trans-1,2-Diphenyloxirane
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula empírica (Notação de Hill):
C14H12O
Número CAS:
Peso molecular:
196.24
Beilstein:
82740
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Ensaio
98%
pf
65-67 °C (lit.)
cadeia de caracteres SMILES
O1[C@@H]([C@H]1c2ccccc2)c3ccccc3
InChI
1S/C14H12O/c1-3-7-11(8-4-1)13-14(15-13)12-9-5-2-6-10-12/h1-10,13-14H/t13-,14-/m1/s1
chave InChI
ARCJQKUWGAZPFX-ZIAGYGMSSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Descrição geral
Trans-stilbene oxide also known as trans-1,2-Diphenyloxirane, is often used in photochemistry, whereit can change its structure when exposed to light.Stilbene oxides can break apart when they are excited bylight, leading to the formation of carbonyl ylides. It is also commonlyused to produce trans-stilbene sulfides.
Aplicação
- Chiral Stationary Phases for Liquid Chromatography: Trans-stilbene oxide has been utilized in the fabrication of cellulose derivative-coated spherical covalent organic frameworks, serving as chiral stationary phases for high-performance liquid chromatographic enantioseparation, demonstrating its pivotal role in advanced analytical methodologies (Yan et al., 2022).
- Method Selection for Chiral High-Performance Liquid Chromatography: Its application extends to the utilization of hysteresis phenomena for chiral high-performance liquid chromatographic method selection in polar organic mode, enhancing the efficiency and specificity of pharmaceutical compound analysis (Horváth et al., 2020).
- Adsorption Properties for Enantioseparations: The effect of chiral selector loading on the adsorption properties of fully- and superficially-porous particles is crucial for high-efficient ultrafast enantioseparations, where trans-stilbene oxide derivatives play a significant role (Felletti et al., 2018).
- Catalysis in Alkene Epoxidation: Trans-stilbene oxide is involved in innovative catalysis research, specifically in the development of carbon nitride-supported Fe(2) cluster catalysts for alkene epoxidation, showcasing its utility in sustainable chemical synthesis (Tian et al., 2018).
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Neil Everall et al.
Applied spectroscopy, 64(1), 52-60 (2010-02-06)
Picosecond time-resolved transmission Raman data were acquired for 1 mm thick powder samples of trans-stilbene, and a Monte Carlo model was developed that can successfully model the laser and Raman pulse profiles. Photon migration broadened the incident (approximately 1 ps)
Joel Putnam et al.
Journal of chromatography. A, 1218(31), 5157-5165 (2011-07-06)
Acid/base mobile phase modifiers affect enantioseparations in ways that are not yet understood for the lack of systematic studies, which makes the scale-up of preparative separations difficult to predict. Shifts of the selectivity of certain pairs of enantiomers upon exposure
A L Slitt et al.
Drug metabolism and disposition: the biological fate of chemicals, 34(7), 1190-1197 (2006-04-20)
trans-Stilbene oxide (TSO) is a synthetic proestrogen that induces phase I and II drug-metabolizing enzymes in rat liver. The purpose of this study was to determine whether TSO also induces transporter expression in rat liver and whether gene induction in
V J Mayani et al.
Chirality, 21(2), 255-261 (2008-06-19)
Chromatographic behavior of nonracemic mixtures, viz., mandelic acid and stilbene oxide as analytes has been studied in detailed by enantiomer self-disproportionation on achiral ordered mesoporous material M41S and regular silica gel as stationary phases. Enantiomer self-disproportionation gave enhanced separation of
Li-Ming Yang et al.
Bioorganic & medicinal chemistry letters, 12(7), 1013-1015 (2002-03-23)
A new series of trans-stilbene benzenesulfonamide derivatives were designed and synthesized as potential antitumor agents. These new compounds were evaluated in the National Cancer Institute's 60 human tumor cell line in vitro screen. Compounds 9-13 were cytotoxic against several cell
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica