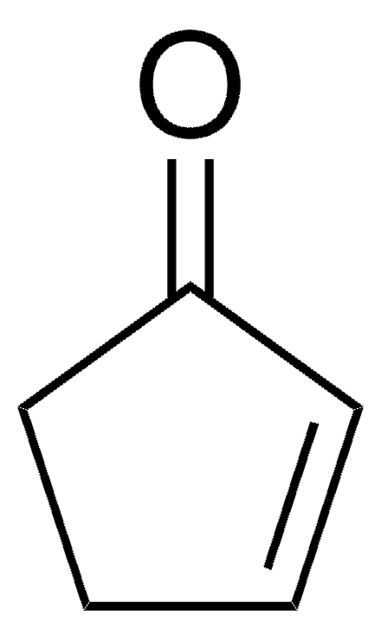
E51309
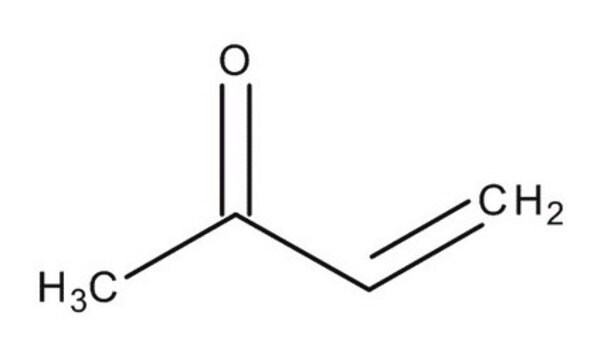
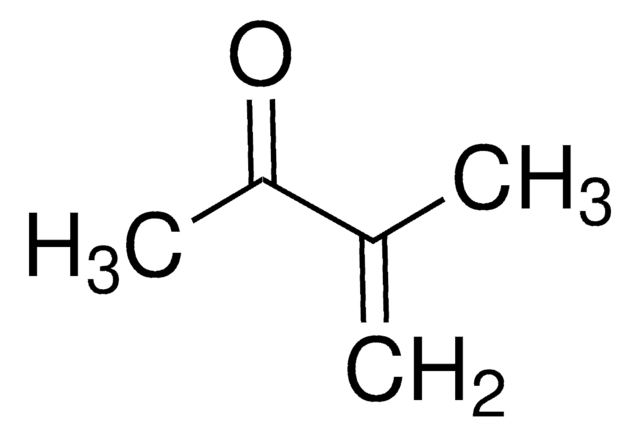
1-Penten-3-one
contains 0.1% BHT as stabilizer, 97%
Sinônimo(s):
Ethyl vinyl ketone
About This Item
Produtos recomendados
Nível de qualidade
Ensaio
97%
Formulário
liquid
contém
0.1% BHT as stabilizer
índice de refração
n20/D 1.419 (lit.)
p.e.
38 °C/60 mmHg (lit.)
densidade
0.851 g/mL at 20 °C
0.845 g/mL at 25 °C (lit.)
temperatura de armazenamento
−20°C
cadeia de caracteres SMILES
CCC(=O)C=C
InChI
1S/C5H8O/c1-3-5(6)4-2/h3H,1,4H2,2H3
chave InChI
JLIDVCMBCGBIEY-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
Aplicação
- Conjugated dienes via palladium-catalyzed Heck reaction with vinyl bromides.
- β-Amino carbonyl derivatives via solvent-free aza-Michael addition reaction with aromatic amines in the presence of ionic liquid catalyst.
- α-Exo-methylene group bearing β-amino carbonyl compounds via ion-supported triphenylphosphine catalyzed aza‐Morita‐Baylis-Hillman reaction with N-tosyl arylimines.
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Eye Dam. 1 - Flam. Liq. 2 - Skin Corr. 1B
Código de classe de armazenamento
3 - Flammable liquids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
14.0 °F - closed cup
Ponto de fulgor (°C)
-10 °C - closed cup
Equipamento de proteção individual
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica