D194255
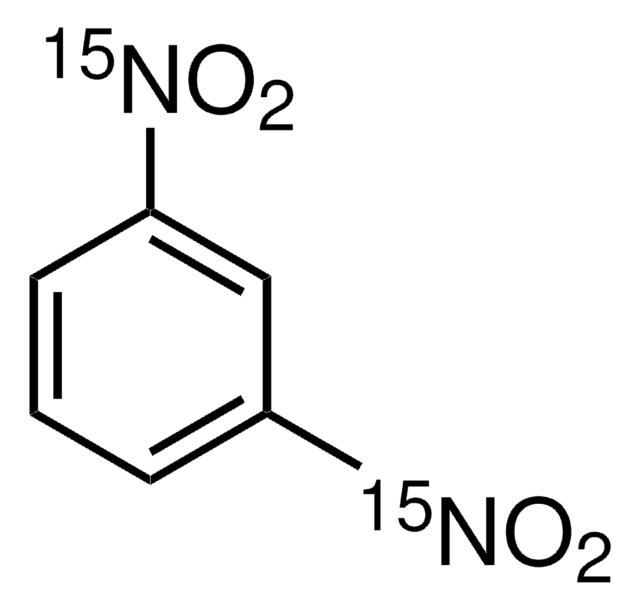
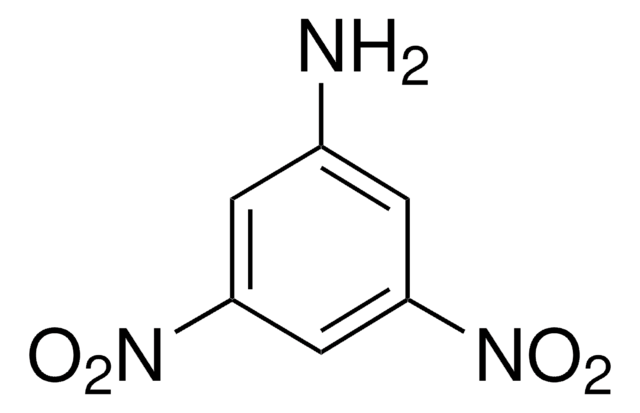
1,3-Dinitrobenzene
97% anhydrous basis
Sinônimo(s):
1,3-Dinitrobenzene, DNB, Meta-dinitrobenzene, m-Dinitrobenzene
About This Item
Produtos recomendados
Nível de qualidade
Ensaio
97% anhydrous basis
Formulário
solid
Impurezas
≤10.0% H2O
p.e.
297 °C (lit.)
pf
84-86 °C (lit.)
densidade
1.368 g/mL at 25 °C (lit.)
cadeia de caracteres SMILES
[O-][N+](=O)c1cccc(c1)[N+]([O-])=O
InChI
1S/C6H4N2O4/c9-7(10)5-2-1-3-6(4-5)8(11)12/h1-4H
chave InChI
WDCYWAQPCXBPJA-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
Aplicação
- 2′,6′- dinitrobiphenyl-4-ol and 1-nitrodibenzofuran via copper-catalyzed regioselective cross-coupling reaction with 4-iodophenol and 2-iodophenol respectively.
- 1H-indazole derivatives by reacting with various N-tosylhydrazones in the presence of a base catalyst.
- 3-(1H-Tetrazol-1-yl)benzenamine by reacting with triethyl orthoformate and NaN3 in the presence of a three-functional redox catalytic system.
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Acute Tox. 1 Dermal - Acute Tox. 2 Inhalation - Acute Tox. 2 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - STOT RE 2
Código de classe de armazenamento
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
302.0 °F - closed cup
Ponto de fulgor (°C)
150 °C - closed cup
Equipamento de proteção individual
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| D194255-25G | 4061835150243 |
| D194255-100G | 4061833561126 |
| D194255-5G |
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica