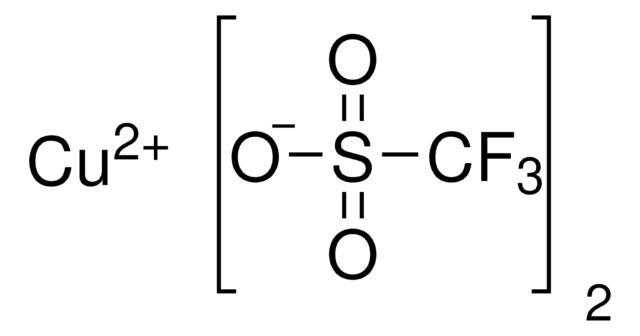796549
Stahl Aerobic Oxidation TEMPO solution
0.2 M in acetonitrile, Solution for Oxidation of Primary Alcohols
About This Item
Produtos recomendados
Formulário
liquid
adequação da reação
reagent type: oxidant
concentração
0.2 M in acetonitrile
temperatura de armazenamento
2-8°C
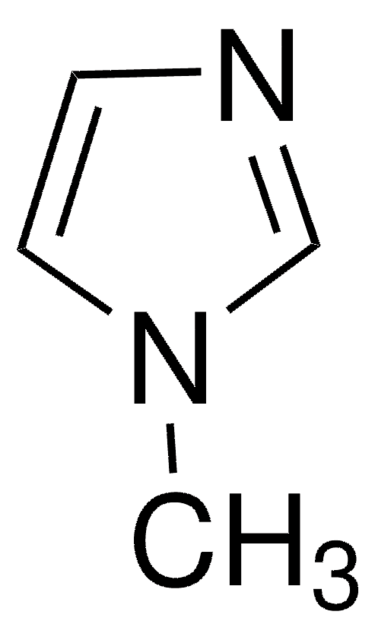
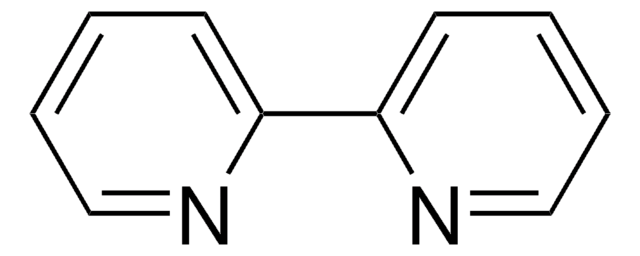
cadeia de caracteres SMILES
CN1C=CN=C1.CC2(C)CCCC(C)(C)N2[O].C3(C4=NC=CC=C4)=NC=CC=C3
InChI
1S/C10H8N2.C9H18NO.C4H6N2/c1-3-7-11-9(5-1)10-6-2-4-8-12-10;1-8(2)6-5-7-9(3,4)10(8)11;1-6-3-2-5-4-6/h1-8H;5-7H2,1-4H3;2-4H,1H3
chave InChI
BQFURWVGIDXRNB-UHFFFAOYSA-N
Descrição geral
Aplicação
produto relacionado
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Dam. 1 - Flam. Liq. 2 - Repr. 2 - Skin Corr. 1C
Código de classe de armazenamento
3 - Flammable liquids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
35.6 °F
Ponto de fulgor (°C)
2.0 °C
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Artigos
Alcohol oxidation is one of the most frequently performed oxidation reactions in organic chemistry. The aldehyde and ketone products of alcohol oxidation are useful intermediates en route to complex molecules.
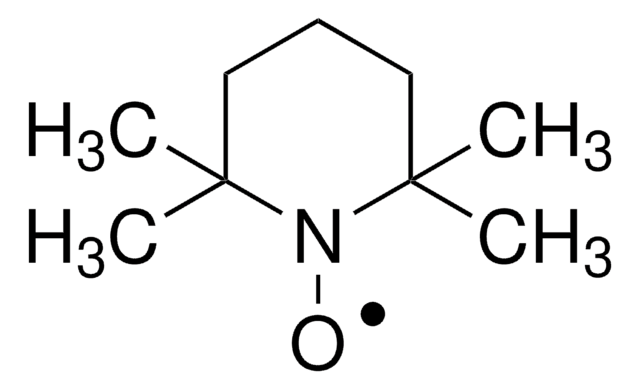
TEMPO (2,2,6,6-Tetramethylpiperidinyloxy or 2,2,6,6-Tetramethylpiperidine 1-oxyl) and its derivatives are stable nitroxy radicals used as catalysts in organic oxidation reactions. TEMPO was discovered by Lebedev and Kazarnovskii in 1960. The stable free radical nature of TEMPO is due to the presence of bulky substituent groups, which hinder the reaction of the free radical with other molecules.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica
![9-Azabicyclo[3.3.1]nonane N-oxyl 95%](/deepweb/assets/sigmaaldrich/product/structures/287/155/e2f4a2e1-1d4e-4bed-9187-9e16d23cbbbf/640/e2f4a2e1-1d4e-4bed-9187-9e16d23cbbbf.png)
![1,8-Diazabiciclo[5,4,0]undec-7-eno 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)