693049
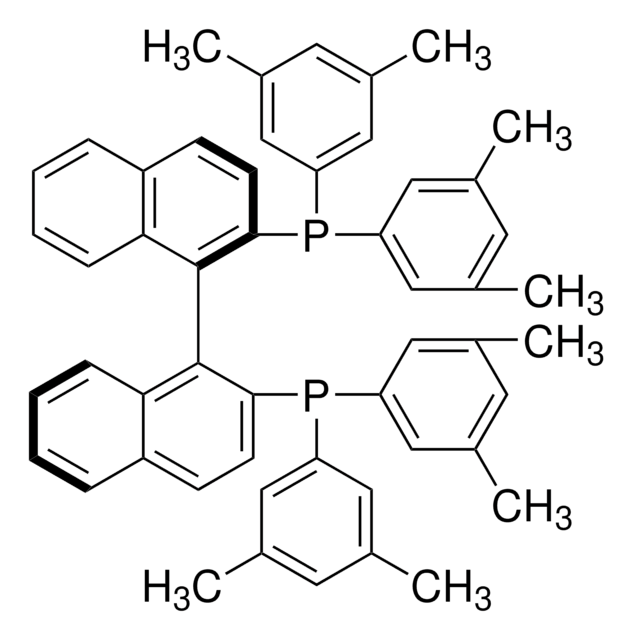
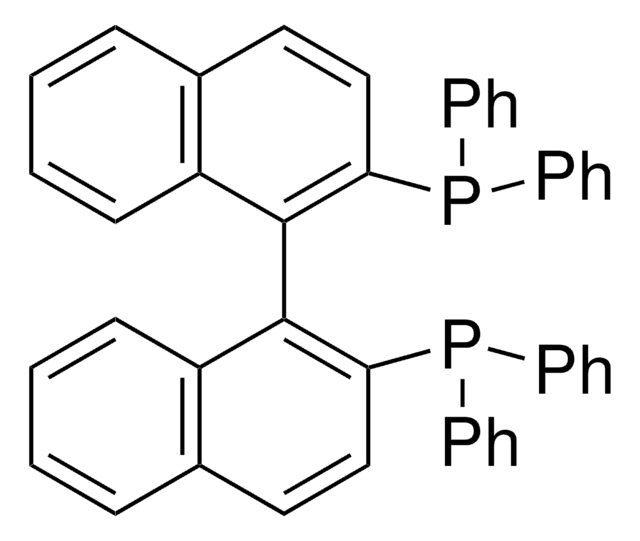
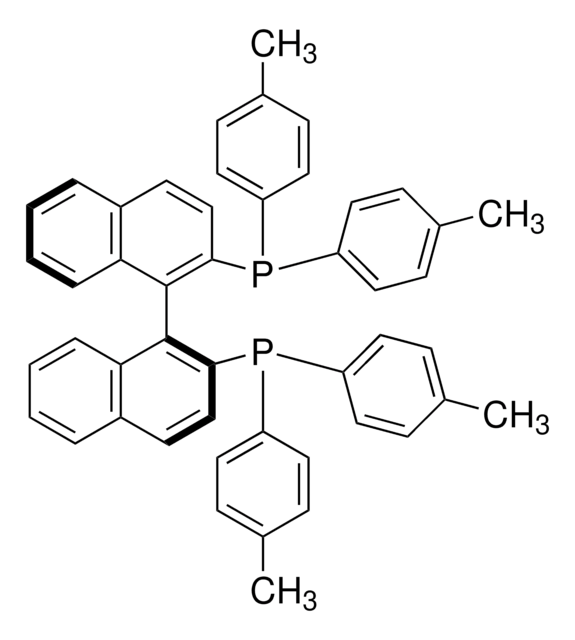
(R)-Tol-BINAP
Sinônimo(s):
(R)-(+)-2,2′-Bis(di-p-tolylphosphino)-1,1′-binaphthyl
About This Item
Produtos recomendados
Formulário
solid
Nível de qualidade
atividade óptica
[α]20/D +162°, c = 0.5 in benzene
adequação da reação
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: ligand
reaction type: Cross Couplings
pf
254-258 °C
grupo funcional
phosphine
cadeia de caracteres SMILES
Cc1ccc(cc1)P(c2ccc(C)cc2)c3ccc4ccccc4c3-c5c(ccc6ccccc56)P(c7ccc(C)cc7)c8ccc(C)cc8
InChI
1S/C48H40P2/c1-33-13-23-39(24-14-33)49(40-25-15-34(2)16-26-40)45-31-21-37-9-5-7-11-43(37)47(45)48-44-12-8-6-10-38(44)22-32-46(48)50(41-27-17-35(3)18-28-41)42-29-19-36(4)20-30-42/h5-32H,1-4H3
chave InChI
IOPQYDKQISFMJI-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
Aplicação
Reactant serving as a precursor for:
- Catalysts used for reductive amination of ketones
- Rh(I)-catalyst for hydrogenation of acetamidoacrylic acid derivatives
- Chiral platinum catalysts for asymmetric Baeyer-Villiger oxidation of cyclic ketones
- CuI-Tol-BINAP catalysts for enantioselective Michael reactions of Grignard reagents to unsaturated esters
- BINAP Pt Dications for cation trapping
Informações legais
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Artigos
The Baeyer-Villiger oxidation is the oxidative cleavage of a carbon-carbon bond adjacent to a carbonyl, which converts the ketones to esters and the cyclic ketones to lactones.
We present an article concerning BINAP/SEGPHOS® Ligands and Complexes.
Hydrogenation, Asymmetric Catalysis, Binap, SEGPHOS®, Aldol reaction, Alkenylation, Arylation, Mannich reaction, Fluorination, Michael addition, Hydrosilylation, Cycloaddition, Takasago
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica