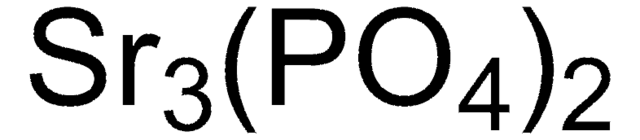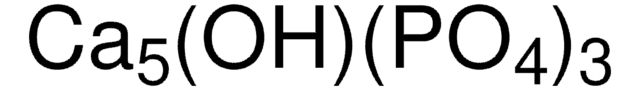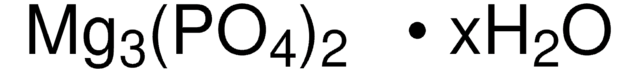677396
Magnesium aluminate, spinel
nanopowder, <50 nm particle size (BET)
Sinônimo(s):
Magnesium aluminate
About This Item
Produtos recomendados
Formulário
nanopowder
adequação da reação
reagent type: catalyst
core: magnesium
área da superfície
>30 m2/g
tamanho de partícula
<50 nm (BET)
pf
2130 °C (lit.)
densidade
3.64 g/mL at 25 °C (lit.)
cadeia de caracteres SMILES
[Mg++].[O-][Al]=O.[O-][Al]=O
InChI
1S/2Al.Mg.4O/q;;+2;;;2*-1
chave InChI
VPBIQXABTCDMAU-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Aplicação
- A Review on Processing Polycrystalline Magnesium Aluminate Spinel (MgAl2O4): This review discusses various sintering techniques, material properties, and machinability of polycrystalline magnesium aluminate spinel, highlighting its optical transparency and mechanical properties (Z Shi et al., 2020).
- Effect of Structure and Composition of Non-Stoichiometry Magnesium Aluminate Spinel on Water Adsorption: This study examines how the structure and composition of magnesium aluminate spinel affect its water adsorption capabilities, focusing on the interaction with gases at different spinel compositions (Y Mordekovitz et al., 2020).
- Spectroscopic Study of Ordering in Non-Stoichiometric Magnesium Aluminate Spinel: The paper explores the ordering within non-stoichiometric magnesium aluminate spinel and its impact on material properties (V Erukhimovitch et al., 2015).
- Transparent Polycrystalline Magnesium Aluminate Spinel Fabricated by Spark Plasma Sintering: This research presents the fabrication of transparent polycrystalline magnesium aluminate spinel using spark plasma sintering, analyzing its mechanical and optical properties (M Sokol et al., 2018).
- A Comparative Study on the Effect of Different Additives on the Formation and Densification of Magnesium Aluminate Spinel: Investigates the effects of various additives on the formation and densification of magnesium aluminate spinel, providing insights into the synthesis and optimization of this ceramic material (SK Mohan et al., 2016).
Informações legais
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órgãos-alvo
Respiratory system
Código de classe de armazenamento
13 - Non Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica