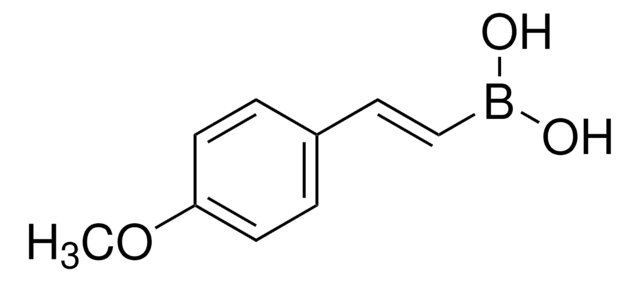
417599
4-Methoxyphenylboronic acid
≥95.0%
Sinônimo(s):
(4-Methoxyphenyl)boric acid, (p-Methoxyphenyl)boronic acid, 4-Anisylboronic acid, 4-Methoxybenzeneboronic acid, p-Anisylboronic acid, p-Methoxybenzeneboronic acid
About This Item
Produtos recomendados
Nível de qualidade
Ensaio
≥95.0%
Formulário
powder
pf
204-206 °C (lit.)
cadeia de caracteres SMILES
COc1ccc(cc1)B(O)O
InChI
1S/C7H9BO3/c1-11-7-4-2-6(3-5-7)8(9)10/h2-5,9-10H,1H3
chave InChI
VOAAEKKFGLPLLU-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Aplicação
- Suzuki-Miyaura cross-coupling reactions
- Pd-catalyzed direct arylation
- Highly effective synthesis using palladium-catalyzed arylation Suzuki-Miyaura cross-coupling in water
- Palladium-catalyzed stereoselective Heck-type reaction
- Tandem-type Pd(II)-catalyzed oxidative Heck reaction and intramolecular C-H amidation sequence
- Copper-mediated ligandless aerobic fluoroalkylation of arylboronic acids with fluoroalkyl iodides
- Ruthenium catalyzed direct arylation
- Rh-catalyzed asymmetric conjugate addition
- Ligand-free copper-catalyzed coupling
Reagent used in Preparation of
- Palladium(II) thiocarboxamide complexes as Suzuki coupling catalyst
- Push-pull arylvinyldiazine chromophores with photophysical properties
Outras notas
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órgãos-alvo
Respiratory system
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
dust mask type N95 (US), Eyeshields, Gloves
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 417599-5G | 4061832090412 |
| 417599-1G | 4061832090399 |
| 417599-25G | 4061832090405 |
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica
![[Pd(OAc)2]3 reagent grade, 98%](/deepweb/assets/sigmaaldrich/product/structures/508/249/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f/640/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f.png)