375217
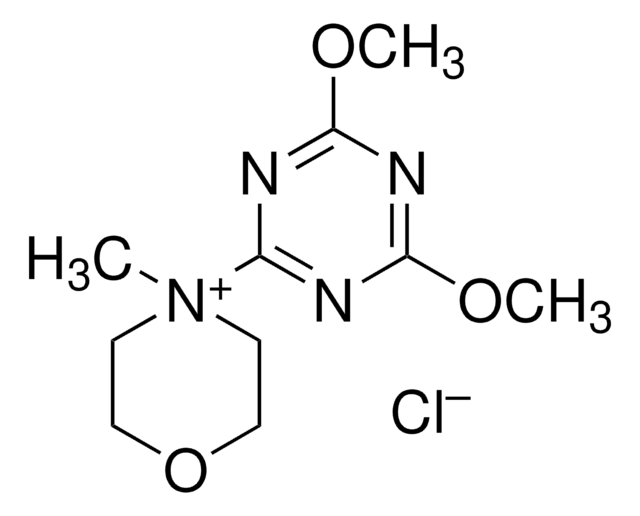
2-Chloro-4,6-dimethoxy-1,3,5-triazine
97%
Sinônimo(s):
2,4-Dimethoxy-6-chloro-1,3,5-triazine, 2,4-Dimethoxy-6-chloro-s-triazine, 2-Chloro-4,6-dimethoxy-1,3,5-triazine, 2-Chloro-4,6-dimethoxy-s-triazine, 4,6-Dimethoxy-2-chloro-s-triazine, 6-Chloro-2,4-dimethoxy-s-triazine, CDMT, Chlorodimethoxytriazine
About This Item
Produtos recomendados
Nível de qualidade
Ensaio
97%
Formulário
solid
pf
71-74 °C (lit.)
grupo funcional
chloro
cadeia de caracteres SMILES
COc1nc(Cl)nc(OC)n1
InChI
1S/C5H6ClN3O2/c1-10-4-7-3(6)8-5(9-4)11-2/h1-2H3
chave InChI
GPIQOFWTZXXOOV-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
Aplicação
- The specific labeling of streptavidin by the modular method for affinity labeling (MoAL).
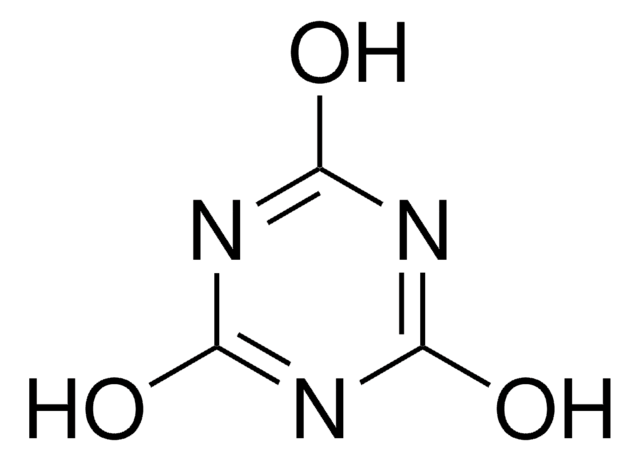
- The preparation of bis(4,6-dimethoxy-1,3,5-triazin-2-yl) ether, via reaction with 2-hydroxy-4,6-dimethoxy-1,3,5-triazine.
- The preparation of 2-(4,6-dimethoxy-1,3,5-triazinyl)trialkylammonium salts, via reaction with various tertiary amines.
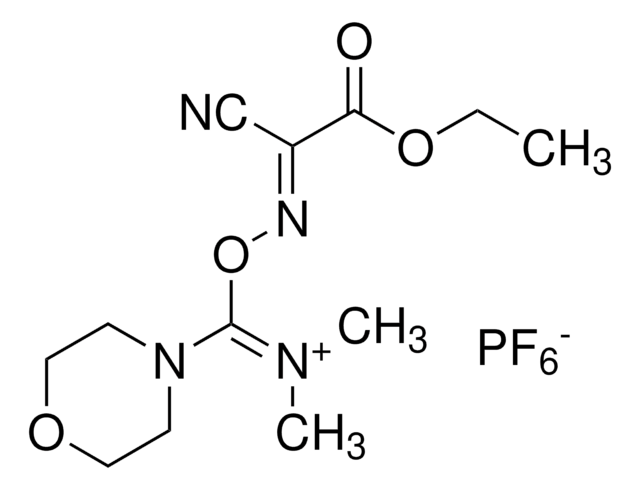
- The synthesis of 4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholinium chloride, via coupling with N-methylmorpholine in THF.
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Aquatic Chronic 2 - Eye Dam. 1 - Skin Corr. 1B - Skin Sens. 1
Código de classe de armazenamento
8A - Combustible corrosive hazardous materials
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
dust mask type N95 (US), Eyeshields, Gloves
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 375217-25G | |
| 375217-1G | |
| 375217-5G | 4061831834963 |
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica