About This Item
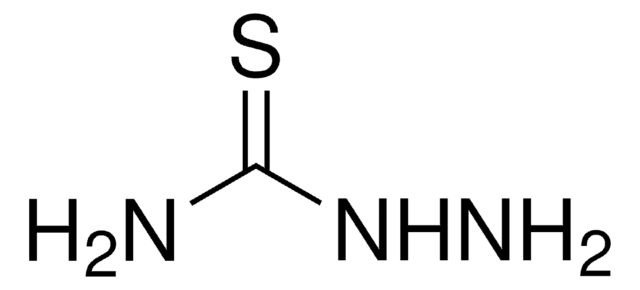
Fórmula linear:
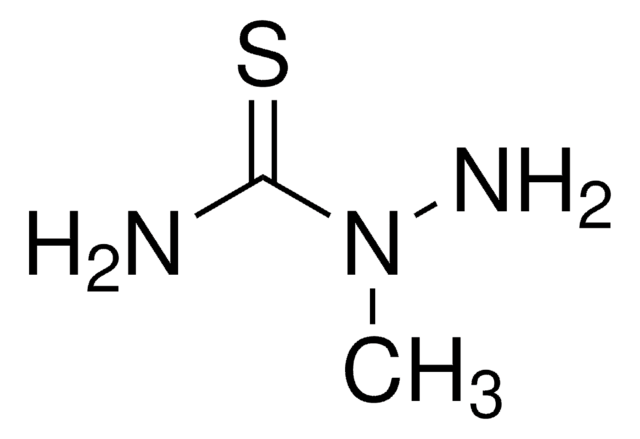
H2NNHCSN(CH3)2
Número CAS:
Peso molecular:
119.19
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Nível de qualidade
Ensaio
98%
pf
153 °C (dec.) (lit.)
grupo funcional
amine
hydrazine
thiourea
temperatura de armazenamento
2-8°C
cadeia de caracteres SMILES
CN(C)C(=S)NN
InChI
1S/C3H9N3S/c1-6(2)3(7)5-4/h4H2,1-2H3,(H,5,7)
chave InChI
FCPHVJQWZFNNKD-UHFFFAOYSA-N
Descrição geral
4,4-Dimethyl-3-thiosemicarbazide is alkyl derivative of thiosemicarbazide. Structure of 4,4-dimethyl-3-thiosemicarbazide in solution has been investigated by NMR and in solid state by IR and X-ray crystallographic methods.
Aplicação
4,4-Dimethyl-3-thiosemicarbazide may be employed as chemical additive to investigate the corrosion inhibition of mild steel in acidic conditions.
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Acute Tox. 4 Oral
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Structures of anti, Z-4, 4-dimethyl-3-thiosemicarbazide, syn, E, Z-2, 4-dimethyl-3-thiosemicarbazide and syn, E-1-cyclopentano-3-thiosemicarbazone.
Valente EJ, et al.
Journal of Chemical Crystallography, 28(1), 27-33 (1998)
Molecular dynamics and quantum chemical calculation studies on 4, 4-dimethyl-3-thiosemicarbazide as corrosion inhibitor in 2.5 MH< sub> 2</sub> SO< sub> 4</sub>.
Musa AY, et al.
Materials Chemistry and Physics, 129(1), 660-665 (2011)
Fang Xie et al.
Journal of biological inorganic chemistry : JBIC : a publication of the Society of Biological Inorganic Chemistry (2018-07-15)
Copper 8-hydroxyquinoline-2-carboxaldehyde-thiosemicarbazide complex (CuHQTS) is a copper complex with strong anticancer activity against cisplatin-resistant neuroblastoma and prostate cancer cells in vitro by cell proliferation assay or fluorescent microscopic imaging. This study aimed to evaluate anti-prostate cancer activity of CuHQTS in
Liang Tian et al.
Biosensors & bioelectronics, 110, 110-117 (2018-04-01)
Enzyme mimics have been developed for bioassay of nucleic acids, with some of them involving complicated labeling. Herein, we report a label-free bioassay for ultrasensitive electronic determination of microRNA at an ultralow concentration based on target-triggered long-range self-assembly DNA-based hybridization
Franco Bisceglie et al.
Metallomics : integrated biometal science, 8(12), 1255-1265 (2016-11-15)
A comparative study between two bisthiosemicarbazones, 2,3-butanedione bis(4,4-dimethyl-3-thiosemicarbazone) and 2,3-butanedione bis(2-methyl-3-thiosemicarbazone), and their copper(ii) complexes is reported. The four compounds have been tested on a leukemia cell line U937 (p53-null) and on an adenocarcinoma cell line A549. The study includes
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica