349631
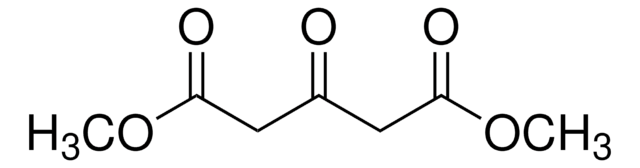
Dimethyl 2-oxoglutarate
96%
Sinônimo(s):
Dimethyl α-ketoglutarate
About This Item
Produtos recomendados
Ensaio
96%
Formulário
liquid
índice de refração
n20/D 1.439 (lit.)
p.e.
90-95 °C/0.4 mmHg (lit.)
densidade
1.203 g/mL at 25 °C (lit.)
grupo funcional
ester
ketone
cadeia de caracteres SMILES
COC(=O)CCC(=O)C(=O)OC
InChI
1S/C7H10O5/c1-11-6(9)4-3-5(8)7(10)12-2/h3-4H2,1-2H3
chave InChI
TXIXSLPEABAEHP-UHFFFAOYSA-N
Categorias relacionadas
Descrição geral
Aplicação
Código de classe de armazenamento
10 - Combustible liquids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Artigos
Sigma-Aldrich presents an article about how proliferatively active cells require both a source of carbon and of nitrogen for the synthesis of macromolecules. Although a large proportion of tumor cells utilize aerobic glycolysis and shunt metabolites away from mitochondrial oxidative phosphorylation, many tumor cells exhibit increased mitochondrial activity.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica

![1,4-Diazabicyclo[2.2.2]octane ReagentPlus®, ≥99%](/deepweb/assets/sigmaaldrich/product/structures/366/129/a6ff4175-974d-4fac-9038-b35e508ef252/640/a6ff4175-974d-4fac-9038-b35e508ef252.png)


![1,8-Diazabiciclo[5,4,0]undec-7-eno 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)