19240
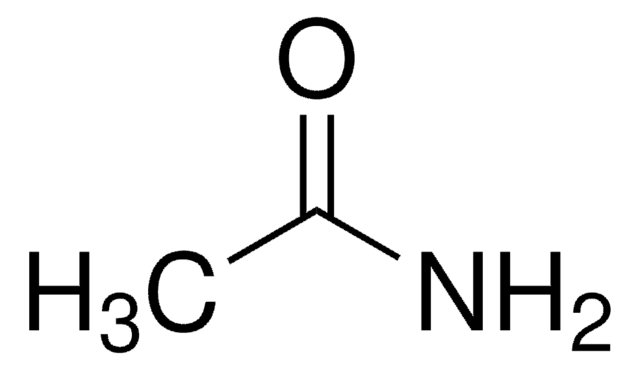
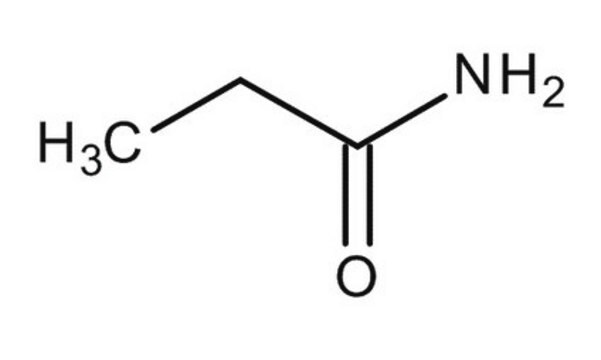
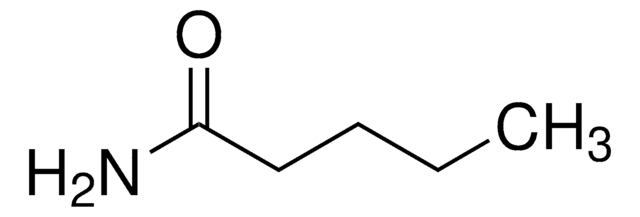
Butyramide
≥98.0% (T)
Sinônimo(s):
Amide C4
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula linear:
CH3CH2CH2CONH2
Número CAS:
Peso molecular:
87.12
Beilstein:
1361528
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Ensaio
≥98.0% (T)
pf
114-116 °C
solubilidade
alcohol: soluble(lit.)
diethyl ether: slightly soluble(lit.)
water: soluble(lit.)
grupo funcional
amide
cadeia de caracteres SMILES
CCCC(N)=O
InChI
1S/C4H9NO/c1-2-3-4(5)6/h2-3H2,1H3,(H2,5,6)
chave InChI
DNSISZSEWVHGLH-UHFFFAOYSA-N
Informações sobre genes
rat ... Ggt1(116568)
Procurando produtos similares? Visita Guia de comparação de produtos
Aplicação
Butyramide was used in the synthesis of hydroxamic acids, electrorheological fluids and β-amodoorganotin compounds. It was used as substrate of (+)-γ-lactamase to develop a microreactor to study enzyme stability, activity, kinetics and substrate specificity.
Código de classe de armazenamento
13 - Non Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
dust mask type N95 (US), Eyeshields, Gloves
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Hongjian Zhang et al.
Drug metabolism and disposition: the biological fate of chemicals, 35(5), 795-805 (2007-02-17)
2-{Butyryl-[2'-(4,5-dimethyl-isoxazol-3-ylsulfamoyl)-biphenyl-4-ylmethyl]-amino}-N-isopropyl-3-methyl-butyramide (BMS-1) is a potent dual acting angiotensin-1 and endothelin-A receptor antagonist. The compound was subject to rapid metabolic clearance in monkey and human liver microsomes and exhibited low systemic exposure and marked interanimal variability in cynomolgus monkeys after p.o.
Effect of butyrate analogues on proliferation and differentiation in human neuroblastoma cell lines.
P Rocchi et al.
Anticancer research, 18(2A), 1099-1103 (1998-06-06)
Butyric acid has been shown in vitro to produce cytodifferentiation of a wide variety of neoplastic cells. The potential clinical use of this compound as a therapeutic agent is limited by its rapid metabolism. This has led to the examination
B P O'Hara et al.
Protein engineering, 13(2), 129-132 (2000-03-10)
The AmiC protein in Pseudomonas aeruginosa is the negative regulator and ligand receptor for an amide-inducible aliphatic amidase operon. In the wild-type PAC1 strain, amidase expression is induced by acetamide or lactamide, but not by butyramide. A mutant strain of
Rajendra Singh et al.
Bioprocess and biosystems engineering, 41(8), 1225-1232 (2018-05-12)
Butyramide is a commodity chemical having wide range of applications from material science to biological sciences including synthesis of therapeutic drugs, hydroxamic acids, and electrorheological fluids. The nitrile hydratase protein of Bacillus sp. APB-6 was explored to develop an efficient
R A Norman et al.
The Journal of biological chemistry, 275(39), 30660-30667 (2000-07-13)
Expression of the amidase operon of Pseudomonas aeruginosa is controlled by AmiC, the ligand sensor and negative regulator, and AmiR the transcription antitermination factor activator. We have titrated out AmiC repression activity in vivo by increased AmiR production in trans
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica