M7571
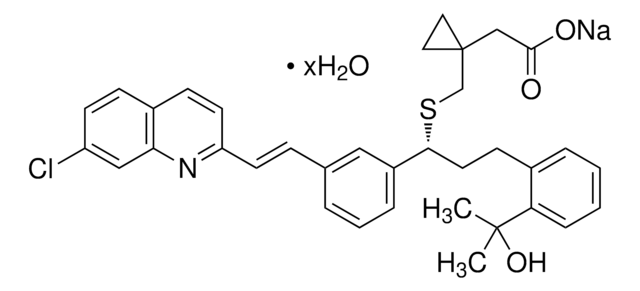
MK-571 sodium salt hydrate
≥95% (HPLC), powder, leukotriene D4 antagonist
Synonym(s):
5-(3-(2-(7-Chloroquinolin-2-yl)ethenyl)phenyl)-8-dimethylcarbamyl-4,6-dithiaoctanoic acid sodium salt hydrate, L-660711
About This Item
Recommended Products
Product Name
MK-571 sodium salt hydrate, ≥95% (HPLC)
Quality Level
assay
≥95% (HPLC)
form
powder
storage condition
desiccated
color
white to beige
solubility
H2O: 15 mg/mL, clear
originator
Merck & Co., Inc., Kenilworth, NJ, U.S.
shipped in
wet ice
storage temp.
−20°C
SMILES string
O.[Na+].CN(C)C(=O)CCSC(SCCC([O-])=O)c1cccc(\C=C\c2ccc3ccc(Cl)cc3n2)c1
InChI
1S/C26H27ClN2O3S2.Na.H2O/c1-29(2)24(30)12-14-33-26(34-15-13-25(31)32)20-5-3-4-18(16-20)6-10-22-11-8-19-7-9-21(27)17-23(19)28-22;;/h3-11,16-17,26H,12-15H2,1-2H3,(H,31,32);;1H2/q;+1;/p-1/b10-6+;;
InChI key
MSHRPLRGSQECLY-DOLBFOAYSA-M
Application
- as an efflux inhibitor for monitoring multidrug resistance protein (MRP)-function and to avoid redundancy of other transporters
- to assess its effect on cell proliferation and 2D-migration in vitro in various cell lines of glioblastoma multiforme (GBM)
- as multidrug resistance (MDR) transporter inhibitor to study its effects in ovarian cancer cells
- as specific inhibitors of ABCC1/2 to investigate transport, toxicity, flow cytometry and arsenic efflux
Biochem/physiol Actions
Features and Benefits
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Sigma-Aldrich offers many products related to leukotriene receptors for your research needs.
Related Content
Discover Bioactive Small Molecules for Lipid Signaling Research
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service