T4424
Trypsin solution from porcine pancreas
1 ×, sterile-filtered, BioReagent, suitable for cell culture, 2.5 g porcine trypsin per liter in Hanks′ Balanced Salt Solution with phenol red
About This Item
Recommended Products
biological source
Porcine pancreas
Quality Level
sterility
sterile-filtered
product line
BioReagent
form
solution
mol wt
23.4 kDa
concentration
1 ×
technique(s)
cell culture | mammalian: suitable
impurities
Porcine parvovirus, none detected (9 CFR)
pH
7.0-7.6
shipped in
dry ice
storage temp.
−20°C
Looking for similar products? Visit Product Comparison Guide
Application
For trypsin digestion of peptides, use a ratio of about 1:100 to 1:20 for trypsin:peptide. The typical use for this product is in removing adherent cells from a culture surface. The concentration of trypsin necessary to dislodge cells from their substrate is dependent primarily on the cell type and the age of the culture. Trypsins have also been used for the re-suspension of cells during cell culture, in proteomics research for digestion of proteins and in various in-gel digestions. Additional applications include assessing crystallization by membrane-based techniques and in a study to determine that protein folding rates and yields can be limited by the presence of kinetic traps.
Biochem/physiol Actions
Serine protease inhibitors, including DFP, TLCK, APMSF, AEBSEF, and aprotinin, amongst others, will inhibit Trypsin.
Caution
Unit Definition
Preparation Note
Storage Class
12 - Non Combustible Liquids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
Trypsin may be used to remove adherent cells from a culture surface. Cells are most commonly removed from the culture substrate by treatment with trypsin or trypsin/EDTA solutions.
Related Content
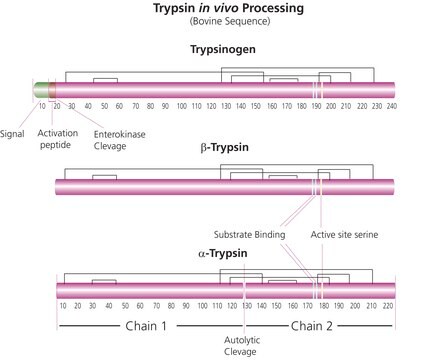
Trypsin is an enzyme in the serine protease class that consists of a polypeptide chain of 223 amino acid residues. Multiple sources, grades and formulations of trypsin specifically designed for research applications are available.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service