All Photos(2)
About This Item
Linear Formula:
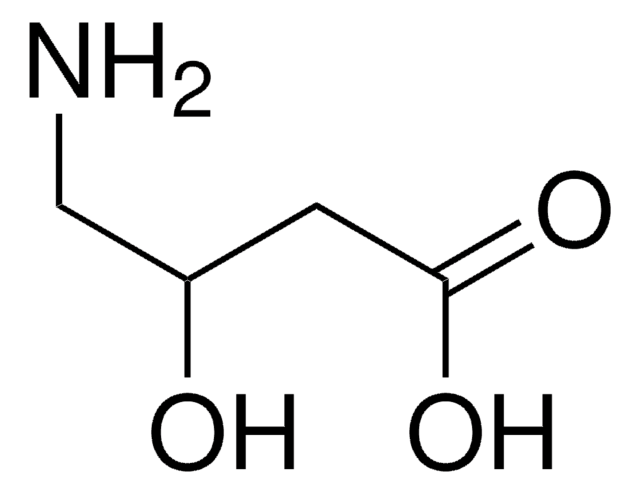
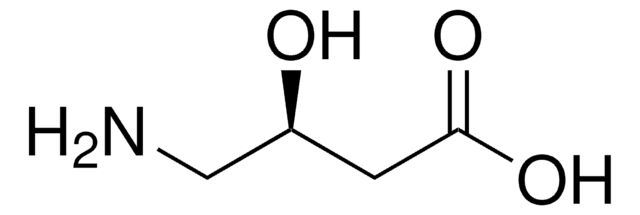
H2NCH2CH2CH(OH)CO2H
CAS Number:
Molecular Weight:
119.12
Beilstein/REAXYS Number:
1721686
MDL number:
UNSPSC Code:
12352116
PubChem Substance ID:
EC Index Number:
670-305-7
NACRES:
NA.22
Recommended Products
Quality Level
assay
96%
optical activity
[α]23/D −30°, c = 1 in H2O
mp
200-203 °C (lit.)
functional group
amine
carboxylic acid
hydroxyl
SMILES string
NCC[C@H](O)C(O)=O
InChI
1S/C4H9NO3/c5-2-1-3(6)4(7)8/h3,6H,1-2,5H2,(H,7,8)/t3-/m0/s1
InChI key
IVUOMFWNDGNLBJ-VKHMYHEASA-N
Looking for similar products? Visit Product Comparison Guide
General description
(S)-(-)-4-Amino-2-hydroxybutyric acid is an important moiety of butirosin, an aminoglycoside antibiotic.
Application
(S)-(-)-4-Amino-2-hydroxybutyric acid may be used in the preparation of 6′-amino-1-N-[(S)-4-amino-2-hydroxybutyryl]-6′-deoxylividomycin A.
Building block for enantiopure 3-hydroxypyrrolidin-2-ones.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Y Ota et al.
The Journal of antibiotics, 53(10), 1158-1167 (2001-01-02)
Butirosin is an interesting 2-deoxystreptamine (DOS)-containing aminoglycoside antibiotic produced by non-actinomycete Bacilli. Recently we were successful in purification of 2-deoxy-scyllo-inosose synthase from butirosin-producer Bacillus circulans as the key enzyme for the biosynthesis of DOS, in cloning of the responsible gene
Bentley, J.M. et al.
Journal of the Chemical Society. Chemical Communications, 231-231 (1995)
M Philippe et al.
The Journal of antibiotics, 35(11), 1507-1512 (1982-11-01)
A semisynthetic aminoglycoside antibiotic 15, containing a cyclic gamma-amino-alpha-hydroxy acid, related to the 1-N-4-amino-2-hydroxybutyric acid (AHBA) side chain of butirosins and amikacin, has been prepared. Conveniently protected 3,2',6'-tris-N-tert-butoxycarbonylgentamicin C1a (12) was condensed with the phtalimido active ester 10 to give
Synthesis of 6'-Amino-1-N-[(S)-4-Amino-2-Hydroxybutyryl]-6'-Deoxylividomycin A.
Watanabe I, et al.
Bulletin of the Chemical Society of Japan, 48(8), 2303-2305 (1975)
Synthesis of (S)-Isoserine.
Miyazawa T, et al.
Agricultural and Biological Chemistry, 40(8), 1651-1652 (1976)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service