1614669
USP
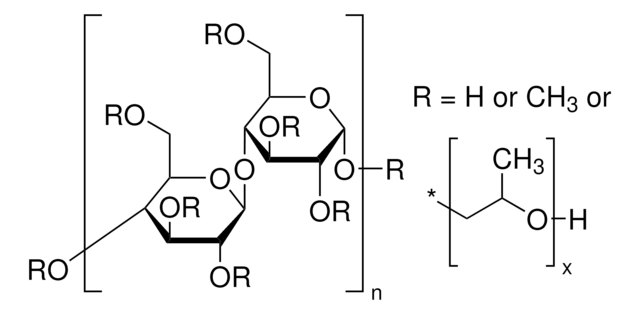
Sodium Starch Glycolate Type A
United States Pharmacopeia (USP) Reference Standard
Zaloguj sięWyświetlanie cen organizacyjnych i kontraktowych
About This Item
Wzór liniowy:
(C2H4O3)x · (Na)x
Numer CAS:
Numer MDL:
Kod UNSPSC:
41116107
NACRES:
NA.24
Polecane produkty
klasa czystości
pharmaceutical primary standard
rodzina API
starch
producent / nazwa handlowa
USP
Zastosowanie
pharmaceutical (small molecule)
Format
neat
Szukasz podobnych produktów? Odwiedź Przewodnik dotyczący porównywania produktów
Opis ogólny
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
Zastosowanie
Wzorzec referencyjny USP glikolanu skrobi sodowej typu A, przeznaczony do stosowania w określonych testach jakości i oznaczeniach, jak określono w kompendiach USP. Również do użytku z monografią USP, taką jak Sodium Starch Glycolate.
Komentarz do analizy
These products are for test and assay use only. They are not meant for administration to humans or animals and cannot be used to diagnose, treat, or cure diseases of any kind.
Inne uwagi
Sales restrictions may apply.
Ta strona może zawierać tekst przetłumaczony maszynowo.
produkt powiązany
Numer produktu
Opis
Cennik
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 1
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Wybierz jedną z najnowszych wersji:
Certyfikaty analizy (CoA)
Lot/Batch Number
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Dokumenty section.
Proszę o kontakt, jeśli potrzebna jest pomoc Obsługa Klienta
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
M Maghsoodi et al.
Pharmaceutical development and technology, 16(3), 243-249 (2010-02-24)
Spherical crystallization (SC) of carbamazepine (CBZ) was carried out for preparation of the agglomerates using the solvent change method. The potential of the intraagglomerate addition of sodium starch glycolate (SSG) as a disintegrant agent and povidone (PVP) as a hydrophilic
Aiman A Obaidat et al.
Acta pharmaceutica (Zagreb, Croatia), 61(1), 83-91 (2011-03-17)
The aim of this study was to prepare fast-dissolving tablets of meloxicam after its complexation with β-cyclodextrin (β-CD) and to investigate the effect of using different superdisintegrants on the disintegration and release of meloxicam from the tablets. A complex of
Sagarika Bose et al.
International journal of pharmaceutics, 393(1-2), 32-40 (2010-02-09)
Film coating is generally accomplished by spraying polymers dissolved in solvents onto a cascading bed of tablets. The limitations associated with the use of solvents (both aqueous and organic) can be overcome by the use of solventless coating technologies. In
Balasubramaniam Jagadish et al.
Chemical & pharmaceutical bulletin, 58(3), 293-300 (2010-03-02)
The present study investigated the effect of co-grinding raloxifene HCL (RHCL) with different superdisintegrants, namely crospovidone (CP), croscarmellose sodium (CCS) and sodium starch glycolate (SSG), using a ball mill, in order to determine the potential effect on dissolution rate and
Ramji Anil Kumar Arza et al.
AAPS PharmSciTech, 10(1), 220-226 (2009-03-12)
Drugs that have narrow absorption window in the gastrointestinal tract (GIT) will have poor absorption. For these drugs, gastroretentive drug delivery systems offer the advantage in prolonging the gastric emptying time. Swellable, floating, and sustained release tablets are developed by
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej